This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Michael O'Shaughnessy/ TS
From Proteopedia
| Line 9: | Line 9: | ||
== Relevance == | == Relevance == | ||
| - | Due to its role in cell division, thymidylate synthase has become a popular target for anticancer drugs. Indirect inhibition of thymidylate synthase by the drug 5-fluorouracil (5-FU) is one of the most used inhibitors for study of TS function. < | + | Due to its role in cell division, thymidylate synthase has become a popular target for anticancer drugs. Indirect inhibition of thymidylate synthase by the drug 5-fluorouracil (5-FU) is one of the most used inhibitors for study of TS function. This drug indirectly inhibits TS as it it eventually converted to FdUMP, which forms a covalent complex with both the active site cysteine and CH<sub>2</sub>H<sub>4</sub>F. |
Revision as of 19:48, 7 April 2022
Contents |
Overview
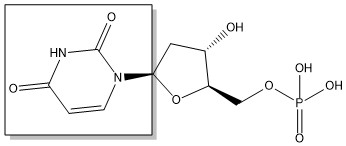
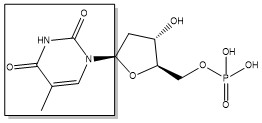
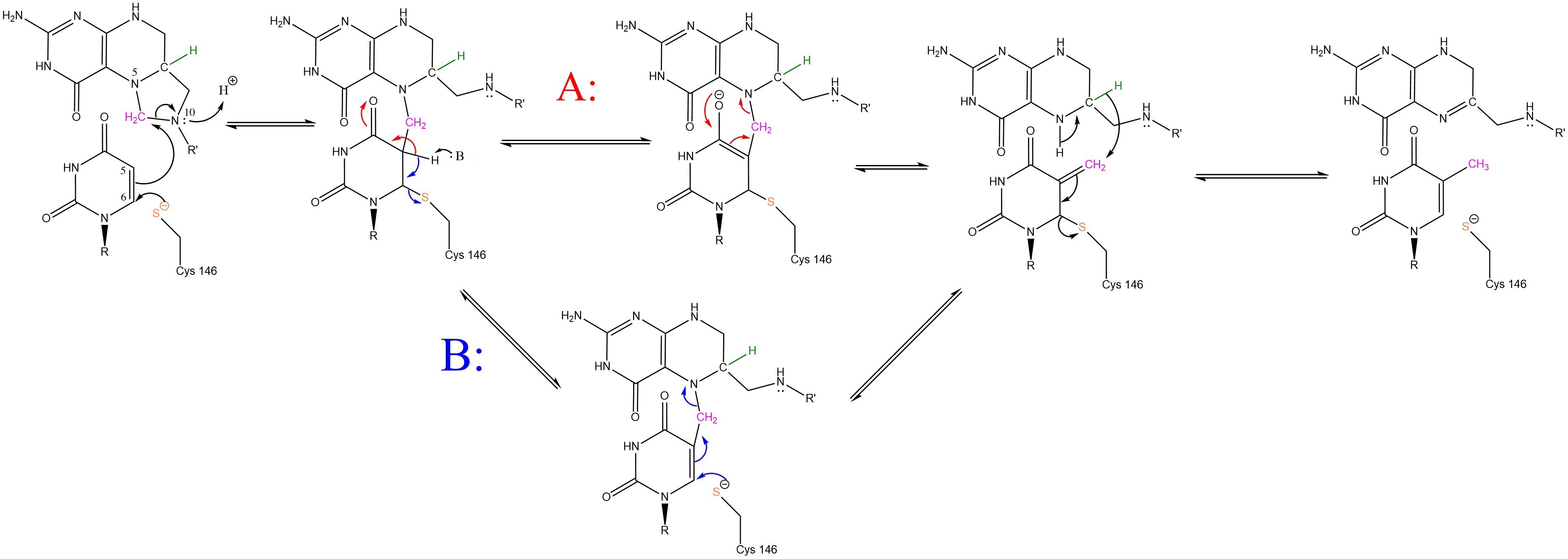
The enzyme Thymidylate Synthase (TS) catalyzes the transfer of a methyl group and a hydride from 5,10-methylenetetrahydrofolate to 2-deoxyuridine-5'-monophosphate, resulting in the formation of thymidine 5'-monophosphate and dihydrofolate. This is the only de novo source of dTMP in humans.
2-deoxyuridine-5'-monophosphate(dUMP) + 5, 10-methylenetetrahydrofolate(CH2H4F) ⇌ thymidine 5'-monophosphate(dTMP) + Dihydrofolate(H2F)
Function
Thymidyalte synthase has a critical role in cell division as the sole producer of thymine in the body.
Disease
Relevance
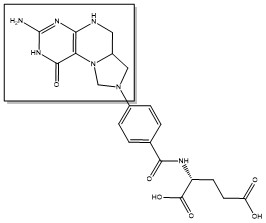
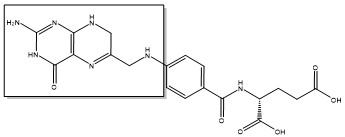
Due to its role in cell division, thymidylate synthase has become a popular target for anticancer drugs. Indirect inhibition of thymidylate synthase by the drug 5-fluorouracil (5-FU) is one of the most used inhibitors for study of TS function. This drug indirectly inhibits TS as it it eventually converted to FdUMP, which forms a covalent complex with both the active site cysteine and CH2H4F.
| |||||||||||
References
- ↑ Hanson, R. M., Prilusky, J., Renjian, Z., Nakane, T. and Sussman, J. L. (2013), JSmol and the Next-Generation Web-Based Representation of 3D Molecular Structure as Applied to Proteopedia. Isr. J. Chem., 53:207-216. doi:http://dx.doi.org/10.1002/ijch.201300024
- ↑ Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644
- ↑ doi: https://dx.doi.org/10.1016/S1074-5521(01)00067-9