This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Kiera Malone/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 88: | Line 88: | ||
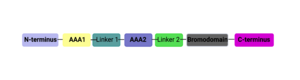
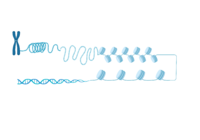
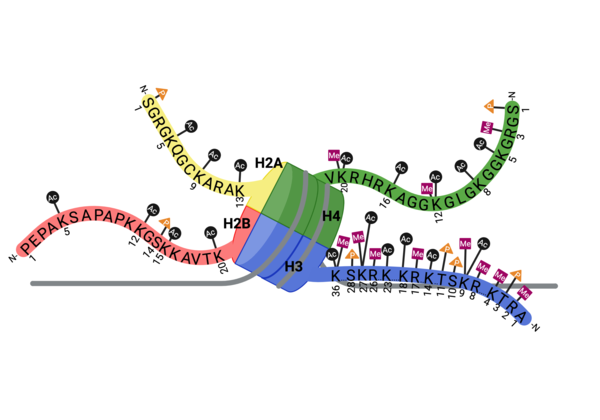
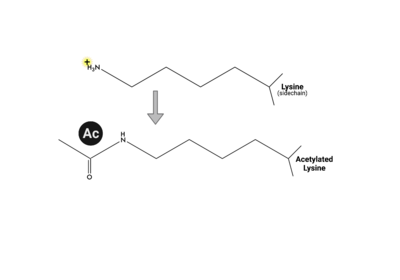
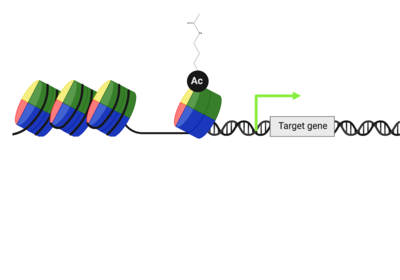
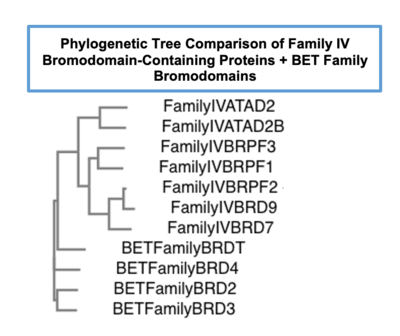
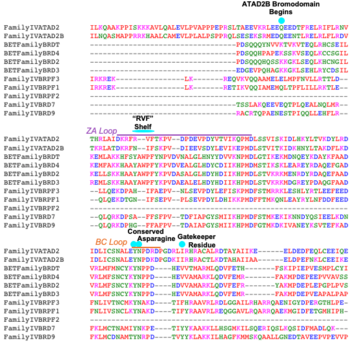
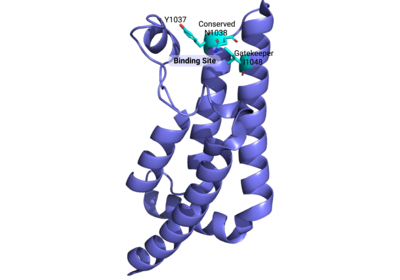
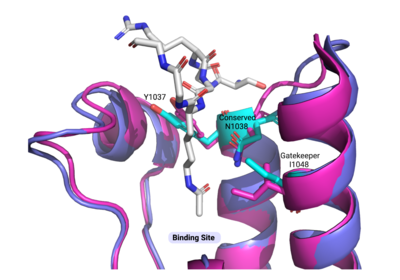
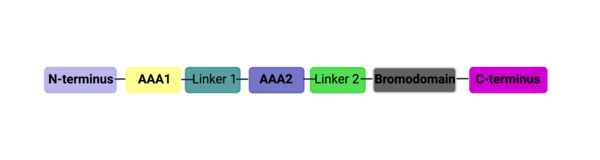
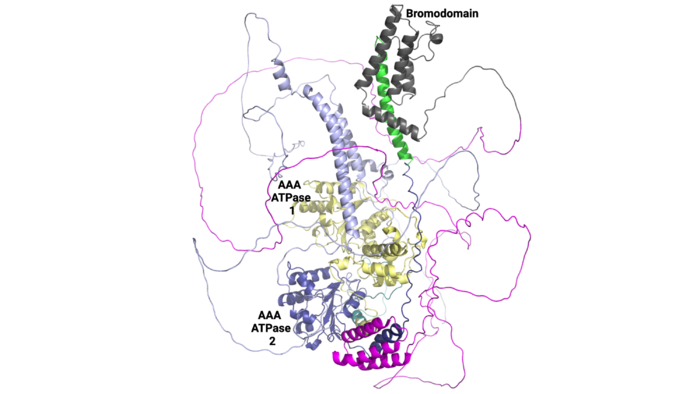
ATAD2 and ATAD2B are chromatin associated proteins (CAPs). Both the ATAD2 and ATAD2B proteins contain two conserved domains that can interact with chromatin: an ''AAA ATPase domain'' and a ''bromodomain''<ref>PMID:22464331</ref>. Additionally, there are linker regions and a C-terminal domain, as represented by the AlphaFold-predicted structure. ATAD2 is a large protein (1,390 amino acids & 158.6 kDa) and has been studied primarily using cellular assays. In these assays, when both AAA ATPases and the bromodomain are present, ATAD2 has a tighter affinity for chromatin, which suggests both are important for the formation of regulatory chromatin complexes(10,4). ATPase domains are known to promote the assembly of coregulator complexes at chromatin, and the ability of the bromodomain to recognize certain histone modifications may specify the location of where ATAD2 is recruited on chromatin(38). Additionally, we know that ATAD2 is recruited to newly synthesized histones on nascent chromatin; moreover, ATAD2 is only present during the S-phase of the cell cycle (KOO), where chromatin remodelers are most active. Chromatin remodeler proteins are usually large complexes, and previously, it was determined that ATAD2 can oligomerize, and the first AAA ATPase is important for this oligomerization (10). Recently, studies of ATAD2 using the yeast homolog ''Abo1'', discovered that the ATPase domains formed a hexamer (using cryo-EM) and was involved with the loading of H3 and H4 onto the NCP. The C-terminal domain was necessary for hexamer stability. However, the bromodomain could not be visualized using cryo-EM. Another yeast homolog, ''Yta7'', found ATAD2 to function as a histone chaperone. The in-depth molecular functions of ATAD2, ''and'' ATAD2B still remain incredible elusive despite recent advances, due to this lack of structural information. | ATAD2 and ATAD2B are chromatin associated proteins (CAPs). Both the ATAD2 and ATAD2B proteins contain two conserved domains that can interact with chromatin: an ''AAA ATPase domain'' and a ''bromodomain''<ref>PMID:22464331</ref>. Additionally, there are linker regions and a C-terminal domain, as represented by the AlphaFold-predicted structure. ATAD2 is a large protein (1,390 amino acids & 158.6 kDa) and has been studied primarily using cellular assays. In these assays, when both AAA ATPases and the bromodomain are present, ATAD2 has a tighter affinity for chromatin, which suggests both are important for the formation of regulatory chromatin complexes(10,4). ATPase domains are known to promote the assembly of coregulator complexes at chromatin, and the ability of the bromodomain to recognize certain histone modifications may specify the location of where ATAD2 is recruited on chromatin(38). Additionally, we know that ATAD2 is recruited to newly synthesized histones on nascent chromatin; moreover, ATAD2 is only present during the S-phase of the cell cycle (KOO), where chromatin remodelers are most active. Chromatin remodeler proteins are usually large complexes, and previously, it was determined that ATAD2 can oligomerize, and the first AAA ATPase is important for this oligomerization (10). Recently, studies of ATAD2 using the yeast homolog ''Abo1'', discovered that the ATPase domains formed a hexamer (using cryo-EM) and was involved with the loading of H3 and H4 onto the NCP. The C-terminal domain was necessary for hexamer stability. However, the bromodomain could not be visualized using cryo-EM. Another yeast homolog, ''Yta7'', found ATAD2 to function as a histone chaperone. The in-depth molecular functions of ATAD2, ''and'' ATAD2B still remain incredible elusive despite recent advances, due to this lack of structural information. | ||
| + | |||
'''ATAD2B has not been extensively studied in the same way as ATAD2, so even less information on structure and function are available.''' | '''ATAD2B has not been extensively studied in the same way as ATAD2, so even less information on structure and function are available.''' | ||
Revision as of 22:31, 30 April 2022
The ATPase Family, AAA Domain-Containing Protein 2B (ATAD2B)
| |||||||||||
References
- ↑ Leachman NT, Brellier F, Ferralli J, Chiquet-Ehrismann R, Tucker RP. ATAD2B is a phylogenetically conserved nuclear protein expressed during neuronal differentiation and tumorigenesis. Dev Growth Differ. 2010 Dec;52(9):747-55. doi: 10.1111/j.1440-169X.2010.01211.x. PMID:21158754 doi:http://dx.doi.org/10.1111/j.1440-169X.2010.01211.x
- ↑ Caron C, Lestrat C, Marsal S, Escoffier E, Curtet S, Virolle V, Barbry P, Debernardi A, Brambilla C, Brambilla E, Rousseaux S, Khochbin S. Functional characterization of ATAD2 as a new cancer/testis factor and a predictor of poor prognosis in breast and lung cancers. Oncogene. 2010 Sep 16;29(37):5171-81. doi: 10.1038/onc.2010.259. Epub 2010 Jun, 28. PMID:20581866 doi:http://dx.doi.org/10.1038/onc.2010.259
- ↑ Kalashnikova EV, Revenko AS, Gemo AT, Andrews NP, Tepper CG, Zou JX, Cardiff RD, Borowsky AD, Chen HW. ANCCA/ATAD2 overexpression identifies breast cancer patients with poor prognosis, acting to drive proliferation and survival of triple-negative cells through control of B-Myb and EZH2. Cancer Res. 2010 Nov 15;70(22):9402-12. doi: 10.1158/0008-5472.CAN-10-1199. Epub , 2010 Sep 23. PMID:20864510 doi:http://dx.doi.org/10.1158/0008-5472.CAN-10-1199
- ↑ Filippakopoulos P, Picaud S, Mangos M, Keates T, Lambert JP, Barsyte-Lovejoy D, Felletar I, Volkmer R, Muller S, Pawson T, Gingras AC, Arrowsmith CH, Knapp S. Histone recognition and large-scale structural analysis of the human bromodomain family. Cell. 2012 Mar 30;149(1):214-31. PMID:22464331 doi:10.1016/j.cell.2012.02.013
- ↑ Mujtaba S, Zeng L, Zhou MM. Structure and acetyl-lysine recognition of the bromodomain. Oncogene. 2007 Aug 13;26(37):5521-7. doi: 10.1038/sj.onc.1210618. PMID:17694091 doi:http://dx.doi.org/10.1038/sj.onc.1210618
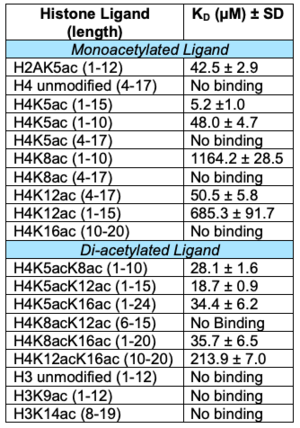
- ↑ Evans CM, Phillips M, Malone KL, Tonelli M, Cornilescu G, Cornilescu C, Holton SJ, Gorjanacz M, Wang L, Carlson S, Gay JC, Nix JC, Demeler B, Markley JL, Glass KC. Coordination of Di-Acetylated Histone Ligands by the ATAD2 Bromodomain. Int J Mol Sci. 2021 Aug 24;22(17). pii: ijms22179128. doi: 10.3390/ijms22179128. PMID:34502039 doi:http://dx.doi.org/10.3390/ijms22179128
- ↑ Lloyd JT, McLaughlin K, Lubula MY, Gay JC, Dest A, Gao C, Phillips M, Tonelli M, Cornilescu G, Marunde MR, Evans CM, Boyson SP, Carlson S, Keogh MC, Markley JL, Frietze S, Glass KC. Structural Insights into the Recognition of Mono- and Diacetylated Histones by the ATAD2B Bromodomain. J Med Chem. 2020 Oct 21. doi: 10.1021/acs.jmedchem.0c01178. PMID:33084328 doi:http://dx.doi.org/10.1021/acs.jmedchem.0c01178
- ↑ Jumper J, Evans R, Pritzel A, Green T, Figurnov M, Ronneberger O, Tunyasuvunakool K, Bates R, Zidek A, Potapenko A, Bridgland A, Meyer C, Kohl SAA, Ballard AJ, Cowie A, Romera-Paredes B, Nikolov S, Jain R, Adler J, Back T, Petersen S, Reiman D, Clancy E, Zielinski M, Steinegger M, Pacholska M, Berghammer T, Bodenstein S, Silver D, Vinyals O, Senior AW, Kavukcuoglu K, Kohli P, Hassabis D. Highly accurate protein structure prediction with AlphaFold. Nature. 2021 Jul 15. pii: 10.1038/s41586-021-03819-2. doi:, 10.1038/s41586-021-03819-2. PMID:34265844 doi:http://dx.doi.org/10.1038/s41586-021-03819-2
- ↑ Varadi M, Anyango S, Deshpande M, Nair S, Natassia C, Yordanova G, Yuan D, Stroe O, Wood G, Laydon A, Zidek A, Green T, Tunyasuvunakool K, Petersen S, Jumper J, Clancy E, Green R, Vora A, Lutfi M, Figurnov M, Cowie A, Hobbs N, Kohli P, Kleywegt G, Birney E, Hassabis D, Velankar S. AlphaFold Protein Structure Database: massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res. 2022 Jan 7;50(D1):D439-D444. doi: 10.1093/nar/gkab1061. PMID:34791371 doi:http://dx.doi.org/10.1093/nar/gkab1061
- ↑ Filippakopoulos P, Picaud S, Mangos M, Keates T, Lambert JP, Barsyte-Lovejoy D, Felletar I, Volkmer R, Muller S, Pawson T, Gingras AC, Arrowsmith CH, Knapp S. Histone recognition and large-scale structural analysis of the human bromodomain family. Cell. 2012 Mar 30;149(1):214-31. PMID:22464331 doi:10.1016/j.cell.2012.02.013