This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Kiera Malone/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
| - | <scene name='90/909366/Color/2'>ATAD2B</scene> is | + | <scene name='90/909366/Color/2'>ATAD2B</scene> is an understudied protein, and therefore very little is known about its overall function. It is a nuclear protein that is a highly sequentially and structurally conserved paralog to ATAD2<ref>PMID:21158754</ref>. It is unknown when the gene duplication event occurred that was able to form both ATAD2 and ATAD2B proteins in evolutionary history, or how ATAD2 and ATA2B interact with each other within a cellular context. ATAD2 is a nuclear co-regulator protein and found to be highly overexpressed in many unrelated forms of cancer<ref>PMID:20581866</ref><ref>PMID:20864510</ref>. ATAD2 overexpression is linked to poor prognosis in these cancer patients. In cancer cell proliferation, ATAD2 is a direct target of E2F, a member of the frequently mutated retinoblastoma protein pathway(2). ATAD2 also acts as a cofactor for the ''Myc'' oncogene, and is engaged in a positive feedback loop to continually amplify the expression of itself and other coregulators in cancer(2). The overall biological function of ATAD2 has yet to be elucidated, but has been implicated in stimulating gene expression, promoting chromatin remodeling, and impacting DNA repair. On the other hand, ATAD2B has not been characterized nearly as much as ATAD2, but it is thought to be involved in neuronal development, and was found overexpresed in multiple human tumors (Leachman, lloyd). |
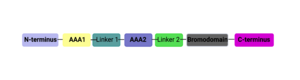
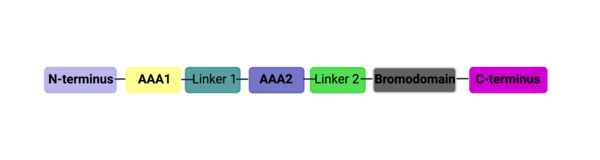
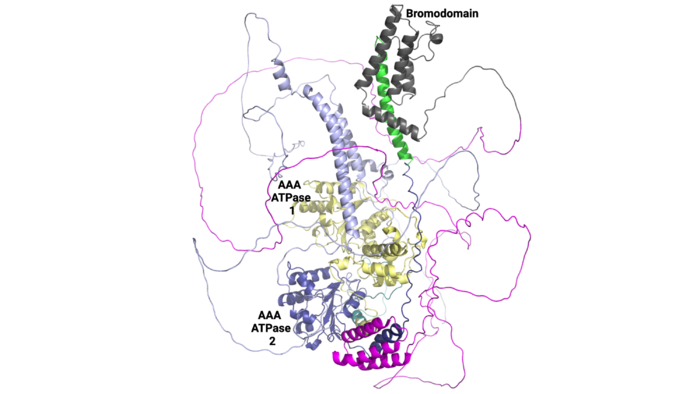
===Structural Organization=== | ===Structural Organization=== | ||
| Line 30: | Line 30: | ||
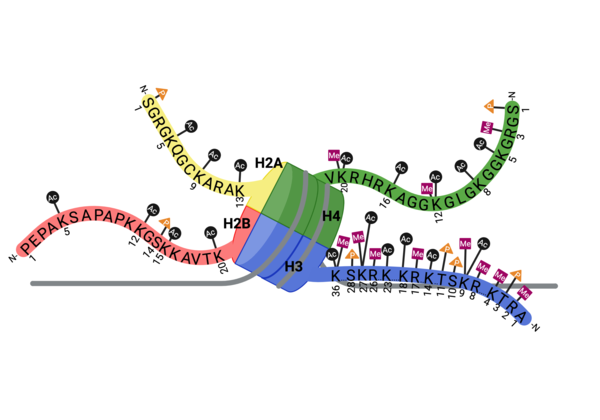
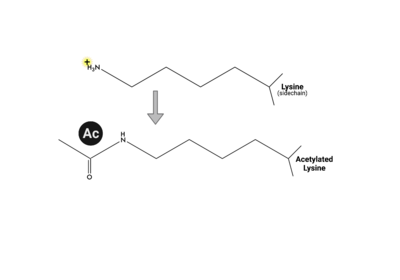
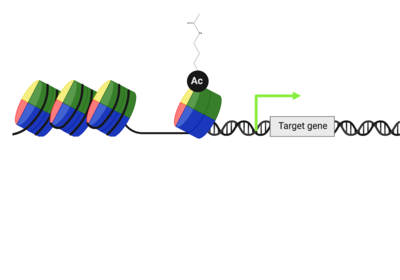
| - | + | Multiple types, numbers, and combinations of PTMs can occur on the histone tails of the nucleosome at any given time. PTMs can serve as docking sites for protein effectors, which impact chromatin structure and gene expression(24,25). Methylation, phosphorylation, and acetylation have been the most well characterized. Methylation modifications provide a platform for regulatory factors associated with gene activation or repression, depending on the modified residue. Phosphorylation ensures proper chromatin compactio during meiosis and mitosis, as well as defining the site of DNA damage. Acetylation neutralizes the positive charge on lysine and arginine residues, loosening histone:DNA packing, for active gene expression. These positively charged lysine residues are found on the tails of histone proteins, and once acetylated, the positive charge the lysine residue carries is removed, which can loosen the DNA compaction around the central octamer of histone proteins and expose a target gene on the DNA make it more amenable to transcription factors. | |
{| | {| | ||
| Line 39: | Line 39: | ||
'''The ''bromodomain'' function is conserved''' | '''The ''bromodomain'' function is conserved''' | ||
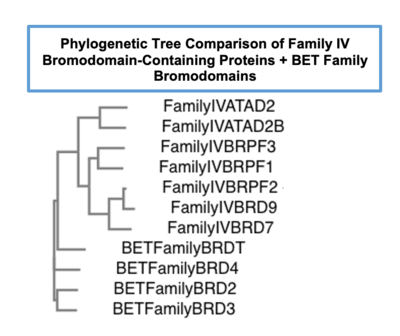
| - | The only known function of the bromodomain is to bind to acetylated lysine residues. This bromodomain function is highly conserved throughout evolution and across 42 bromodomain-containing proteins. Bromodomains have been known about since the 1990s, and in 2012, Filippakopoulos, et al. characterized and divided these bromodomains into eight sub-families based upon similarities in sequence and structure (PF). Despite bromodomains having the same distinct structural fold, the sequences between bromodomains both within and across the different subfamilies, varies greatly. | + | The only known function of the bromodomain is to bind to acetylated lysine residues. This bromodomain function is highly conserved throughout evolution and across 42 bromodomain-containing proteins. Bromodomains can also serve as a site to recruit transcriptional machinery to the target gene once accessible, thus having the power to impact gene expression.Bromodomains have been known about since the 1990s, and in 2012, Filippakopoulos, et al. characterized and divided these bromodomains into eight sub-families based upon similarities in sequence and structure (PF). Despite bromodomains having the same distinct structural fold, the sequences between bromodomains both within and across the different subfamilies, varies greatly. The ATAD2B bromodomain is able to recognize acetylated lysine modifications on histone proteins (see below)(fillikapolous). The bromodomain is the only ATAD2B domain that has been studied, and only little information has been elucidated. |
| - | The ATAD2B bromodomain is able to recognize acetylated lysine modifications on histone proteins (see below)(fillikapolous). The bromodomain is the only ATAD2B domain that has been studied, and only little information has been elucidated. | + | |
{| | {| | ||
| [[Image:FamilyTrees.png|thumb|left|Phylogenetic tree comparing bromodomain-containing proteins in Family IV to the well characterized BET family of bromodomains|400px]] | | [[Image:FamilyTrees.png|thumb|left|Phylogenetic tree comparing bromodomain-containing proteins in Family IV to the well characterized BET family of bromodomains|400px]] | ||
| Line 51: | Line 50: | ||
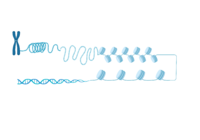
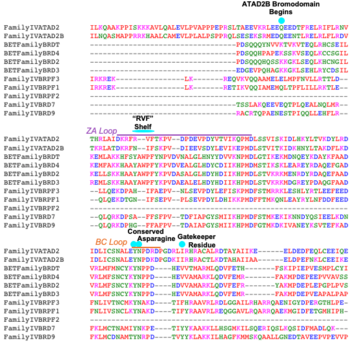
All bromodomain-containing proteins have the same structural motif. It is a left-handed four alpha-helical bundle. These alpha helices are connected by two different and flexible loops whose sequences are variable when compared to the other bromodomain residues, as seen above. These loops are responsible for coordinating the acetylated lysine histone residues to the bromodomain binding pocket. The binding pocket itself is hydrophobic, and for ATAD2 and ATAD2B, deep. | All bromodomain-containing proteins have the same structural motif. It is a left-handed four alpha-helical bundle. These alpha helices are connected by two different and flexible loops whose sequences are variable when compared to the other bromodomain residues, as seen above. These loops are responsible for coordinating the acetylated lysine histone residues to the bromodomain binding pocket. The binding pocket itself is hydrophobic, and for ATAD2 and ATAD2B, deep. | ||
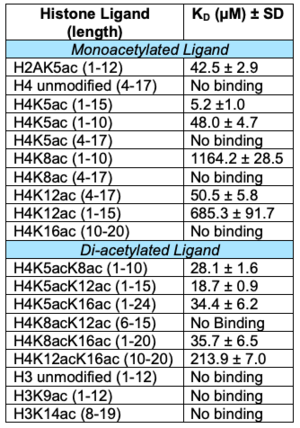
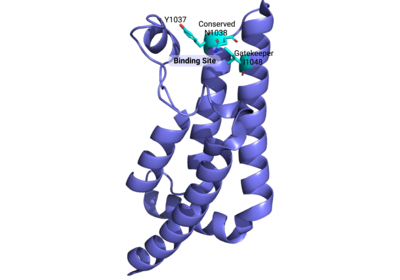
| - | The <scene name='90/909366/Atad2histonebinding/2'>conserved asparagine</scene> is the single most important residue for bromodomain binding to acetylated lysine residues. Other residues that have been conserved throughout evolution, such as the preceding <scene name='90/909366/Atad2histonebinding/3'>tyrosine</scene> residue, are important for binding to the backbone of the histone tail. This works to help stabilize the acetylated lysine residue insertion into the bromodomain binding pocket. Additionally, there is a <scene name='90/909366/Atad2histonebinding/5'>gatekeeper</scene> residue that serves to limit the number of acetylated lysine residues that are inserted into the binding pocket. Another motif in ATAD2 and other bromodomains, termed the "RVF" or "WPF" shelf also helps in ligand specificity in the ZA loop. | + | The <scene name='90/909366/Atad2histonebinding/2'>conserved asparagine</scene> is the single most important residue for bromodomain binding to acetylated lysine residues. Other residues that have been conserved throughout evolution, such as the preceding <scene name='90/909366/Atad2histonebinding/3'>tyrosine</scene> residue, are important for binding to the backbone of the histone tail. This works to help stabilize the acetylated lysine residue insertion into the bromodomain binding pocket. Additionally, there is a hydrophobic <scene name='90/909366/Atad2histonebinding/5'>gatekeeper</scene> residue that serves to limit the number of acetylated lysine residues that are inserted into the binding pocket. Another motif in ATAD2 and other bromodomains, termed the "RVF" or "WPF" shelf also helps in ligand specificity in the ZA loop. ATAD2B has an "NIF" motif, which also helps it to distinguish between different histone ligands in Lloyd, et al. (2020) LLOYD. Interestingly, both ATAD2 and ATAD2B are known to recognize di-acetylated lysine residues on histone tails, but a structure of this interaction has yet to be solved. Unfortunately due to this, we are unable to visualize how both acetylated lysine residues fit in the binding pocket. |
===Bromodomain & ATAD2B Function=== | ===Bromodomain & ATAD2B Function=== | ||
| Line 97: | Line 96: | ||
== Therapeutic Interventions == | == Therapeutic Interventions == | ||
| - | + | Many bromodomain-containing proteins are highly overexpressed in many forms of cancer, and the pharmaceutical industry is keen to design inhibitors to temper bromodomain function in cancer progression. The most well-known inhibitor is a pan-bromodomain inhibitor, JQ1 that was originally designed for the BET Family bromodomains. Over time, it has been characterized to have slightly inhibitory effects in other bromodomains. ATAD2B does not have a specific inhibitor, but ATAD2 has a few currently commercially available. The ATAD2 bromodomain, however, is notoriously extremely difficult to drug, due to the deepness of its binding pocket(16). Only a handful of the compounds developed have been shown to be effective in blocking the binding interaction between ATAD2 and acetylated lysine PTMs (16,18,19). Despite ATAD2B not having its own selective inhibitor, Llloyd, et al. characterized the effects of a known ATAD2 inhibitor, Compound 38, with the ATAD2B bromodomain. | |
| + | |||
| + | ===Compound 38=== | ||
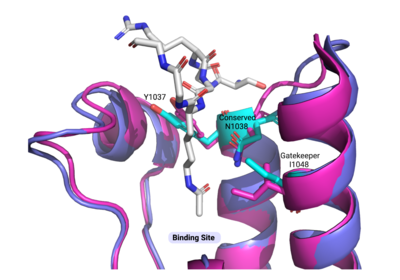
| + | Compound 38 (C-38) was 1.8-fold times more selective for the ATAD2 bromodomain than ATAD2B, with 90 nM and 166.3 nM binding affinities, respectively. They solved the structure of C-38 bound to the ATAD2B bromodomain to determine the specific molecular interactions that are occurring. C-38 was coordinated in the binding pocket much like the acetylated lysine residues of the histone proteins: through hydrogen bonds and hydrophobic interactions. The inhibitor bound to the conserved asparagine (N1038), and was coordinated through hydrophobic interactions with the gatekeeper residue I1048. However, since this compound was manufactured to be specific for the ATAD2 bromodomain, some important interactions between protein and inhibitor are not present in the ATAD2B bromodomain structure, including an important hydrogen bond interaction. These mechanistic molecular differences are thought to play a role in the lower affinity for C-38 with ATAD2B. Overall, however, the coordination of C-38 between ATAD2 and ATAD2B is extremely similar, and Lloyd, et al. postulates that it may be difficult to develop a truly selective inhibitor for each protein. This endeavor is still being explored. | ||
| + | |||
| + | ==Additional Resources== | ||
| + | ATAD2 UniProt Page | ||
| + | ATAD2B UniProt Page | ||
| + | EpiCypher : their technology helps to elucidate the histone interacting partners of bromodomain-containing proteins | ||
| + | |||
| + | |||
| + | ==Acknowledgments== | ||
| + | Glass & Frietze Laboratories : UVM researchers working on learning more about the function of ATAD2, and especially ATAD2B! | ||
</StructureSection> | </StructureSection> | ||
== References == | == References == | ||
<references/> | <references/> | ||
Revision as of 01:25, 1 May 2022
The ATPase Family, AAA Domain-Containing Protein 2B (ATAD2B)
| |||||||||||
References
- ↑ Leachman NT, Brellier F, Ferralli J, Chiquet-Ehrismann R, Tucker RP. ATAD2B is a phylogenetically conserved nuclear protein expressed during neuronal differentiation and tumorigenesis. Dev Growth Differ. 2010 Dec;52(9):747-55. doi: 10.1111/j.1440-169X.2010.01211.x. PMID:21158754 doi:http://dx.doi.org/10.1111/j.1440-169X.2010.01211.x
- ↑ Caron C, Lestrat C, Marsal S, Escoffier E, Curtet S, Virolle V, Barbry P, Debernardi A, Brambilla C, Brambilla E, Rousseaux S, Khochbin S. Functional characterization of ATAD2 as a new cancer/testis factor and a predictor of poor prognosis in breast and lung cancers. Oncogene. 2010 Sep 16;29(37):5171-81. doi: 10.1038/onc.2010.259. Epub 2010 Jun, 28. PMID:20581866 doi:http://dx.doi.org/10.1038/onc.2010.259
- ↑ Kalashnikova EV, Revenko AS, Gemo AT, Andrews NP, Tepper CG, Zou JX, Cardiff RD, Borowsky AD, Chen HW. ANCCA/ATAD2 overexpression identifies breast cancer patients with poor prognosis, acting to drive proliferation and survival of triple-negative cells through control of B-Myb and EZH2. Cancer Res. 2010 Nov 15;70(22):9402-12. doi: 10.1158/0008-5472.CAN-10-1199. Epub , 2010 Sep 23. PMID:20864510 doi:http://dx.doi.org/10.1158/0008-5472.CAN-10-1199
- ↑ Filippakopoulos P, Picaud S, Mangos M, Keates T, Lambert JP, Barsyte-Lovejoy D, Felletar I, Volkmer R, Muller S, Pawson T, Gingras AC, Arrowsmith CH, Knapp S. Histone recognition and large-scale structural analysis of the human bromodomain family. Cell. 2012 Mar 30;149(1):214-31. PMID:22464331 doi:10.1016/j.cell.2012.02.013
- ↑ Mujtaba S, Zeng L, Zhou MM. Structure and acetyl-lysine recognition of the bromodomain. Oncogene. 2007 Aug 13;26(37):5521-7. doi: 10.1038/sj.onc.1210618. PMID:17694091 doi:http://dx.doi.org/10.1038/sj.onc.1210618
- ↑ Evans CM, Phillips M, Malone KL, Tonelli M, Cornilescu G, Cornilescu C, Holton SJ, Gorjanacz M, Wang L, Carlson S, Gay JC, Nix JC, Demeler B, Markley JL, Glass KC. Coordination of Di-Acetylated Histone Ligands by the ATAD2 Bromodomain. Int J Mol Sci. 2021 Aug 24;22(17). pii: ijms22179128. doi: 10.3390/ijms22179128. PMID:34502039 doi:http://dx.doi.org/10.3390/ijms22179128
- ↑ Lloyd JT, McLaughlin K, Lubula MY, Gay JC, Dest A, Gao C, Phillips M, Tonelli M, Cornilescu G, Marunde MR, Evans CM, Boyson SP, Carlson S, Keogh MC, Markley JL, Frietze S, Glass KC. Structural Insights into the Recognition of Mono- and Diacetylated Histones by the ATAD2B Bromodomain. J Med Chem. 2020 Oct 21. doi: 10.1021/acs.jmedchem.0c01178. PMID:33084328 doi:http://dx.doi.org/10.1021/acs.jmedchem.0c01178
- ↑ Jumper J, Evans R, Pritzel A, Green T, Figurnov M, Ronneberger O, Tunyasuvunakool K, Bates R, Zidek A, Potapenko A, Bridgland A, Meyer C, Kohl SAA, Ballard AJ, Cowie A, Romera-Paredes B, Nikolov S, Jain R, Adler J, Back T, Petersen S, Reiman D, Clancy E, Zielinski M, Steinegger M, Pacholska M, Berghammer T, Bodenstein S, Silver D, Vinyals O, Senior AW, Kavukcuoglu K, Kohli P, Hassabis D. Highly accurate protein structure prediction with AlphaFold. Nature. 2021 Jul 15. pii: 10.1038/s41586-021-03819-2. doi:, 10.1038/s41586-021-03819-2. PMID:34265844 doi:http://dx.doi.org/10.1038/s41586-021-03819-2
- ↑ Varadi M, Anyango S, Deshpande M, Nair S, Natassia C, Yordanova G, Yuan D, Stroe O, Wood G, Laydon A, Zidek A, Green T, Tunyasuvunakool K, Petersen S, Jumper J, Clancy E, Green R, Vora A, Lutfi M, Figurnov M, Cowie A, Hobbs N, Kohli P, Kleywegt G, Birney E, Hassabis D, Velankar S. AlphaFold Protein Structure Database: massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res. 2022 Jan 7;50(D1):D439-D444. doi: 10.1093/nar/gkab1061. PMID:34791371 doi:http://dx.doi.org/10.1093/nar/gkab1061
- ↑ Filippakopoulos P, Picaud S, Mangos M, Keates T, Lambert JP, Barsyte-Lovejoy D, Felletar I, Volkmer R, Muller S, Pawson T, Gingras AC, Arrowsmith CH, Knapp S. Histone recognition and large-scale structural analysis of the human bromodomain family. Cell. 2012 Mar 30;149(1):214-31. PMID:22464331 doi:10.1016/j.cell.2012.02.013