This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Gluconeogenesis
From Proteopedia
(Difference between revisions)
| Line 46: | Line 46: | ||
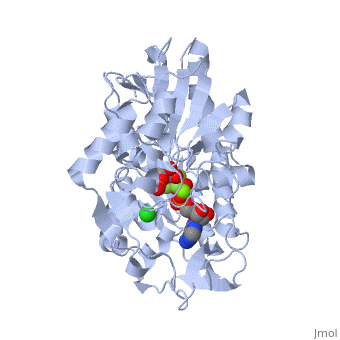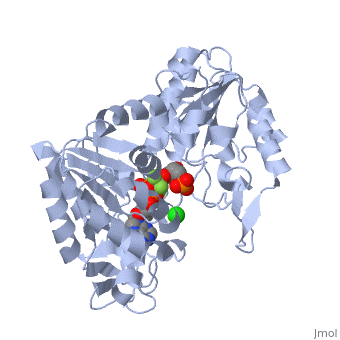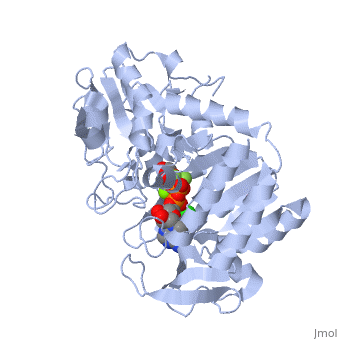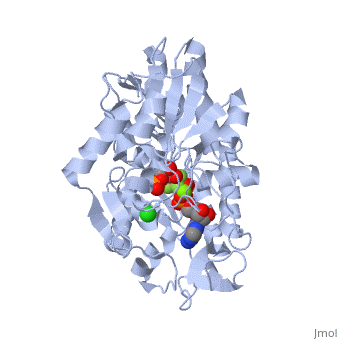
The next steps in the reaction are the same as reversed glycolysis. However, [[fructose 1,6-bisphosphatase]] converts <scene name='39/392339/Cv1/2'>fructose 1,6-bisphosphate</scene> to <scene name='92/925544/Cv/5'>fructose 6-phosphate</scene>, using one water molecule and releasing one phosphate (in glycolysis, phosphofructokinase 1 converts F6P and ATP to F1,6BP and ADP). This is also the rate-limiting step of gluconeogenesis. | The next steps in the reaction are the same as reversed glycolysis. However, [[fructose 1,6-bisphosphatase]] converts <scene name='39/392339/Cv1/2'>fructose 1,6-bisphosphate</scene> to <scene name='92/925544/Cv/5'>fructose 6-phosphate</scene>, using one water molecule and releasing one phosphate (in glycolysis, phosphofructokinase 1 converts F6P and ATP to F1,6BP and ADP). This is also the rate-limiting step of gluconeogenesis. | ||
| + | |||
| + | <scene name='39/392339/Cv/4'>Glucose-6-phosphate</scene> is formed from <scene name='39/392339/Cv1/1'>fructose-6-phosphate</scene> by [[phosphoglucoisomerase]] (the reverse of step 2 in glycolysis). Glucose-6-phosphate can be used in other metabolic pathways or dephosphorylated to free glucose. | ||
</StructureSection> | </StructureSection> | ||
== References == | == References == | ||
<references/> | <references/> | ||
Revision as of 11:26, 6 December 2022
| |||||||||||
References
- ↑ Dunten P, Belunis C, Crowther R, Hollfelder K, Kammlott U, Levin W, Michel H, Ramsey GB, Swain A, Weber D, Wertheimer SJ. Crystal structure of human cytosolic phosphoenolpyruvate carboxykinase reveals a new GTP-binding site. J Mol Biol. 2002 Feb 15;316(2):257-64. PMID:11851336 doi:http://dx.doi.org/10.1006/jmbi.2001.5364