This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1793
From Proteopedia
(Difference between revisions)
| Line 14: | Line 14: | ||
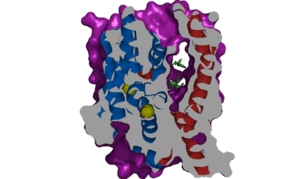
NTCP, among others in the SLC10 family, have <scene name='95/952721/Sodium_binding/2'>two sodium binding sites</scene>. Many polar and positively charged residues are characteristic of these active sites. The high level of conservation among sodium binding placement and interacting residues suggests sodium binding is coupled to bile salt transport. Additional mutations in the X-motif near sodium binding sites have shown that bile salt transport function is lost also suggesting that sodium allows bile salt binding. | NTCP, among others in the SLC10 family, have <scene name='95/952721/Sodium_binding/2'>two sodium binding sites</scene>. Many polar and positively charged residues are characteristic of these active sites. The high level of conservation among sodium binding placement and interacting residues suggests sodium binding is coupled to bile salt transport. Additional mutations in the X-motif near sodium binding sites have shown that bile salt transport function is lost also suggesting that sodium allows bile salt binding. | ||
| - | <Ref name = "Goutam"> Goutam, K., Ielasi, F.S., Pardon, E. et al. Structural basis of sodium-dependent bile salt uptake into the liver. Nature 606, 1015–1020 (2022). [https://doi.org/10.1038/s41586-022-04723-z DOI: 10.1038/s41586-022-04723-z]. </Ref> It is understood that these sodium binding sites facilitate changes from open-pore to closed pore states of NTCP that allow for the binding or release of bile salts. Closed-pore state is favored in the absence of sodium ions, while open-pore state is favored in the presence of sodium ions. This also allows for | + | <Ref name = "Goutam"> Goutam, K., Ielasi, F.S., Pardon, E. et al. Structural basis of sodium-dependent bile salt uptake into the liver. Nature 606, 1015–1020 (2022). [https://doi.org/10.1038/s41586-022-04723-z DOI: 10.1038/s41586-022-04723-z]. </Ref> It is understood that these sodium binding sites facilitate changes from open-pore to closed pore states of NTCP that allow for the binding or release of bile salts. Closed-pore state is favored in the absence of sodium ions, while open-pore state is favored in the presence of sodium ions. This also allows for sodium concentrations to regulate uptake of taurocholates. When intracellular sodium levels are higher, open-pore state is favored allowing for the diffusion of taurocholates. However, when extracellular sodium levels are high, closed-state is favored preventing diffusion of taurocholates. <ref name="Goutam"/> |
Revision as of 00:29, 29 March 2023
| This Sandbox is Reserved from February 27 through August 31, 2023 for use in the course CH462 Biochemistry II taught by R. Jeremy Johnson at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1765 through Sandbox Reserved 1795. |
To get started:
More help: Help:Editing |
Sodium Bile Salt Co-Transporting Protein
| |||||||||||
References
- ↑ 1.0 1.1 Goutam, K., Ielasi, F.S., Pardon, E. et al. Structural basis of sodium-dependent bile salt uptake into the liver. Nature 606, 1015–1020 (2022). DOI: 10.1038/s41586-022-04723-z.