This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1783
From Proteopedia
(Difference between revisions)
| Line 48: | Line 48: | ||
=== HBV/HDV === | === HBV/HDV === | ||
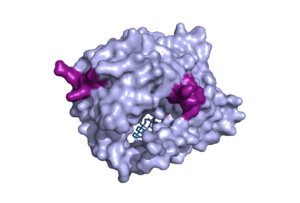
<scene name='95/952711/Pres1_binding_area_on_ntcp/2'>PreS1 Binding</scene> | <scene name='95/952711/Pres1_binding_area_on_ntcp/2'>PreS1 Binding</scene> | ||
| - | Hepatitis B and D both rely on NTCP to bind and infect a human. HBV/HDV uses a similar mechanism as bile salt uptake within NTCP. The myristoylated (myr) pre-S1 domain of HBV is critical for its binding within the protein. Residues 8-17 within the pre-S1 domain were found to be most important in pre-S1 binding to NTCP. HBV/HDV uses the binding patches on the outside of the protein to successfully bind. HBV/HDV binding halts the uptake of bile salt, indicating that the tunnel formed within NTCP allows the uptake of bile salts may also mediate the binding of HBV/HDV[[ | + | Hepatitis B and D both rely on NTCP to bind and infect a human. HBV/HDV uses a similar mechanism as bile salt uptake within NTCP. The myristoylated (myr) pre-S1 domain of HBV is critical for its binding within the protein. Residues 8-17 within the pre-S1 domain were found to be most important in pre-S1 binding to NTCP. HBV/HDV uses the binding patches on the outside of the protein to successfully bind. HBV/HDV binding halts the uptake of bile salt, indicating that the tunnel formed within NTCP allows the uptake of bile salts and may also mediate the binding of HBV/HDV. [[https://www.nature.com/articles/s41586-022-04845-4]] |
</StructureSection> | </StructureSection> | ||
== References == | == References == | ||
<references/> | <references/> | ||
Revision as of 23:33, 30 March 2023
| This Sandbox is Reserved from February 27 through August 31, 2023 for use in the course CH462 Biochemistry II taught by R. Jeremy Johnson at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1765 through Sandbox Reserved 1795. |
To get started:
More help: Help:Editing |
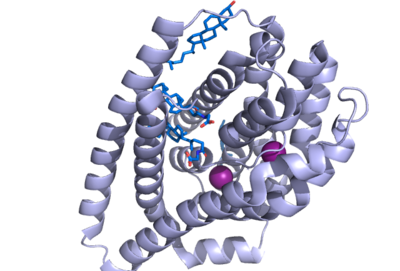
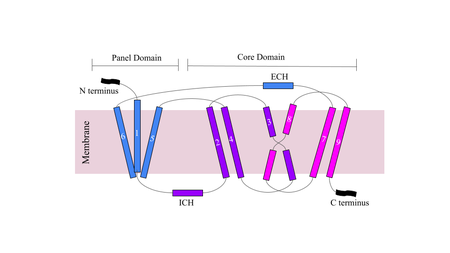
Sodium Taurocholate Co-Transporting Peptide
| |||||||||||
References
- ↑ Goutam, K., Ielasi, F.S., Pardon, E. et al. Structural basis of sodium-dependent bile salt uptake into the liver. Nature 606, 1015–1020 (2022). DOI: 10.1038/s41586-022-04723-z.
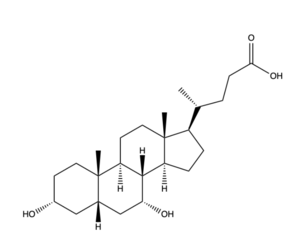
- ↑ Maldonado-Valderrama, J., Wilde, P., Macierzanka, A., & Mackie, A. (2011). The role of bile salts in digestion. Advances in colloid and interface science, 165(1), 36–46. DOI: 10.1016/j.cis.2010.12.002.
- ↑ 3.0 3.1 3.2 Asami J, Kimura KT, Fujita-Fujiharu Y, Ishida H, Zhang Z, Nomura Y, Liu K, Uemura T, Sato Y, Ono M, Yamamoto M, Noda T, Shigematsu H, Drew D, Iwata S, Shimizu T, Nomura N, Ohto U. Structure of the bile acid transporter and HBV receptor NTCP. Nature. 2022 Jun; 606 (7916):1021-1026. DOI: 10.1038/s41586-022-04845-4.
- ↑ Liu, H., Irobalieva, R.N., Bang-Sørensen, R. et al. Structure of human NTCP reveals the basis of recognition and sodium-driven transport of bile salts into the liver. Cell Res 32, 773–776 (2022). DOI: 10.1038/s41422-022-00680-4.