Sandbox Reserved 1794
From Proteopedia
(Difference between revisions)
| Line 27: | Line 27: | ||
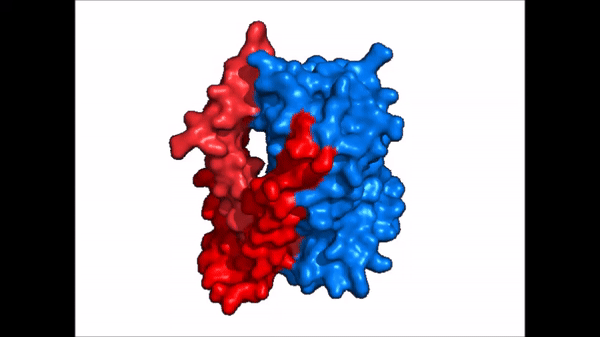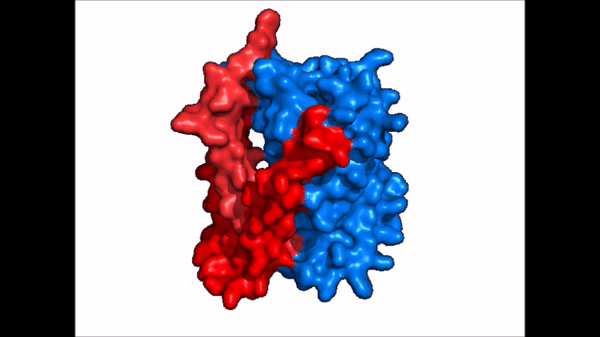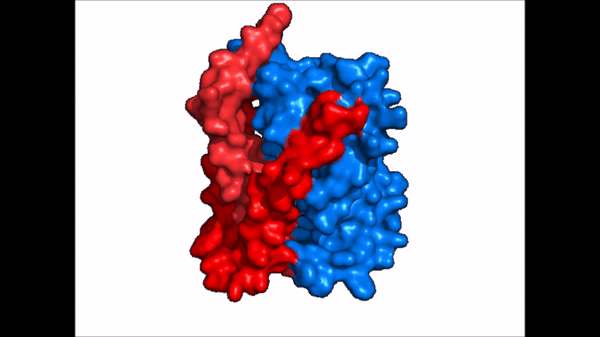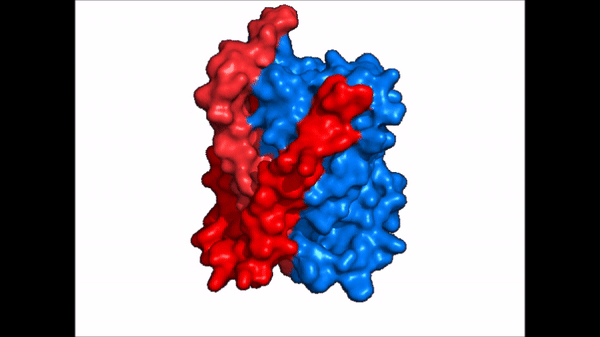
<table align='right' border='0' width='4' cellpadding='0' bgcolor='#d0d0d0' hspace='0'><tr><td rowspan='2'> </td><td bgcolor='#e8e8e8'>[[Image:Surface_NTCP_confchange.gif]]</td></tr><tr><td bgcolor='#e8e8e8'>Cartoon representation of NTCP conformational change.</td></tr></table> | <table align='right' border='0' width='4' cellpadding='0' bgcolor='#d0d0d0' hspace='0'><tr><td rowspan='2'> </td><td bgcolor='#e8e8e8'>[[Image:Surface_NTCP_confchange.gif]]</td></tr><tr><td bgcolor='#e8e8e8'>Cartoon representation of NTCP conformational change.</td></tr></table> | ||
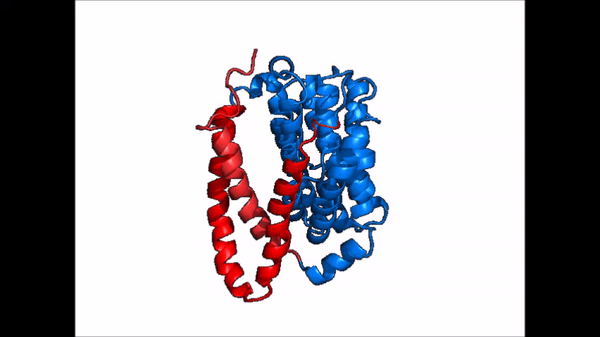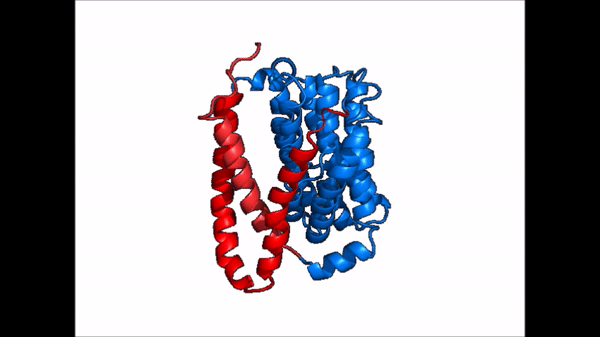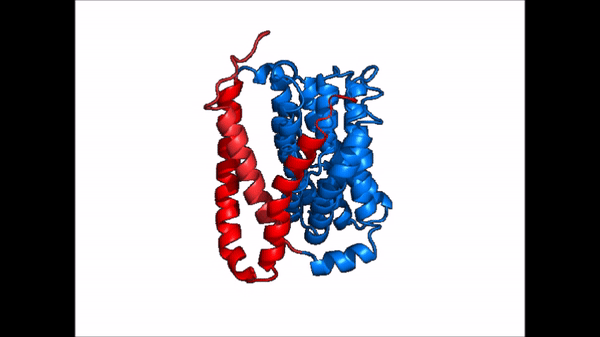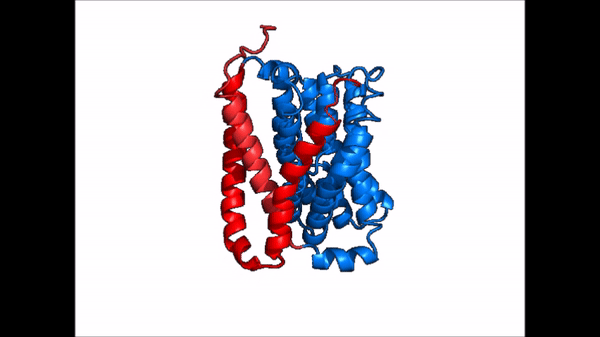
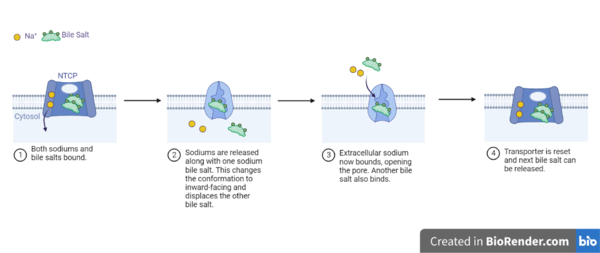
| - | NTCP exists in two different conformations; | + | NTCP exists in two different conformations; the <scene name='95/952722/Ntcp_open_pore/1'>open pore conformation</scene> and the inward-facing conformation. In order to transport bile salts across the plasma membrane of hepocytes, NTCP must undergo the conformational change from inward facing to open pore. This movement consists of the core and panel domains both rotating 20 degrees and the panel domain moving 5 Å away from the core domain, which remains relatively rigid. This conformational change reveals two sodium ion binding sites as well as a pore in the membrane that bile salts can pass through. This movement of the panel domain is facilitated by proline and glycine residues located in the connector loops between the panel and core domains. These residues act as hinges that assist in the movement of the panel domain away from the core domain. |
== Bile Salt Transport == | == Bile Salt Transport == | ||
Revision as of 17:26, 6 April 2023
Sodium Taurocholate Co-Transporting Polypeptide
| |||||||||||
References
- ↑ Stieger B. The role of the sodium-taurocholate cotransporting polypeptide (NTCP) and of the bile salt export pump (BSEP) in physiology and pathophysiology of bile formation. Handb Exp Pharmacol. 2011;(201):205-59. doi: 10.1007/978-3-642-14541-4_5. PMID: 21103971. DOI: DOI: 10.1007/978-3-642-14541-4_5.
- ↑ Geyer, J., Wilke, T. & Petzinger, E. The solute carrier family SLC10: more than a family of bile acid transporters regarding function and phylogenetic relationships. Naunyn Schmied Arch Pharmacol 372, 413–431 (2006). https://doi.org/10.1007/s00210-006-0043-8
- ↑ Park, JH., Iwamoto, M., Yun, JH. et al. Structural insights into the HBV receptor and bile acid transporter NTCP. Nature 606, 1027–1031 (2022). https://doi.org/10.1038/s41586-022-04857-0.
- ↑ 4.0 4.1 Goutam, K., Ielasi, F.S., Pardon, E. et al. Structural basis of sodium-dependent bile salt uptake into the liver. Nature 606, 1015–1020 (2022). DOI: 10.1038/s41586-022-04723-z.
- ↑ Qi X. and Li W. (2022). Unlocking the secrets to human NTCP structure. The Innovation 3(5), 100294. https://doi.org/10.1016/j.xinn.2022.100294
- ↑ 6.0 6.1 Liu, H., Irobalieva, R.N., Bang-Sørensen, R. et al. Structure of human NTCP reveals the basis of recognition and sodium-driven transport of bile salts into the liver. Cell Res 32, 773–776 (2022). https://doi.org/10.1038/s41422-022-00680-4
Student Contributors
- Isabelle White
- Lena Barko