This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1793
From Proteopedia
(Difference between revisions)
| Line 5: | Line 5: | ||
== Introduction == | == Introduction == | ||
| - | + | [[image:Taurocholate.png|thumb|250 px| "Fig. 1 Image of Taurocholic acid a crystalline bile acid"]] | |
| - | NTCP | + | Sodium Taurocholate Co-Transporting Polypeptide, or NTCP, is a [https://en.wikipedia.org/wiki/Membrane_transport_protein membrane transporter protein] that is found in the plasma membrane of liver cells, or [https://en.wikipedia.org/wiki/Hepatocyte hepatocytes]. NTCP's primary function is the transportation of taurocholates, or '''bile salts''', (Fig. 1) into the liver and out of the liver to the small intestine. <Ref> Stieger B. The role of the sodium-taurocholate cotransporting polypeptide (NTCP) and of the bile salt export pump (BSEP) in physiology and pathophysiology of bile formation. Handb Exp Pharmacol. 2011;(201):205-59. doi: 10.1007/978-3-642-14541-4_5. PMID: 21103971. [https://dx.doi.org/10.1007/978-3-642-14541-4_5 DOI: DOI: 10.1007/978-3-642-14541-4_5]. </Ref> Bile salts play various roles in metabolism and digestion, but their main function is the [https://en.wikipedia.org/wiki/Emulsion emulsification] of lipid droplets into smaller fragments. This enables lipases to break down the droplets into their monomers, or triglycerides which are then able to be digested. NTCP is part of the [https://en.wikipedia.org/wiki/Solute_carrier_family solute carrier superfamily], more specifically SLC10. NTCP is the founding member of the SLC10 family, first discovered in rat hepatocytes in 1978. <ref name = "SLC10"> Geyer, J., Wilke, T. & Petzinger, E. The solute carrier family SLC10: more than a family of bile acid transporters regarding function and phylogenetic relationships. Naunyn Schmied Arch Pharmacol 372, 413–431 (2006). https://doi.org/10.1007/s00210-006-0043-8 </ref> NTCP has a key role in [https://en.wikipedia.org/wiki/Enterohepatic_circulation Enterohepatic circulation] or '''bile salt recycling''', and its unique ability to transport other solutes lends it therapeutic potential for lowering cholesterol and treating [https://en.wikipedia.org/wiki/Liver_disease liver disease]. |
| + | NTCP also serves as a binding site for [https://en.wikipedia.org/wiki/Hepatitis_B hepatitis B virus] and [https://en.wikipedia.org/wiki/Hepatitis_D hepatitis D virus]. <ref name = "Park"> Park, JH., Iwamoto, M., Yun, JH. et al. Structural insights into the HBV receptor and bile acid transporter NTCP. Nature 606, 1027–1031 (2022). https://doi.org/10.1038/s41586-022-04857-0. </ref> Future studies into HBV binding mechanism can help understand infection pathways and the development of viral inhibitors. | ||
== Structure == | == Structure == | ||
| Line 19: | Line 20: | ||
==== Bile Salt ==== | ==== Bile Salt ==== | ||
The <scene name='95/952721/Amphipathic_patterns/2'>amphipathic pore</scene> is also characteristic of NTCP. The pore surface remains {{Template:ColorKey_Hydrophobic}}, while lining of the open pore state is largely {{Template:ColorKey_Polar}}. However, in the "inward-facing conformation" the polar pore residues are inaccessible. When the pore is closed only the surface hydrophobic residues are observed. As the pore opens up inner polar residues become accessible allowing for the binding of substrates. <Ref name = The pattern of hydrophobic and polar residues within the pore is believed to follow similar amphipathic patterns within taurocholate and other NTCP substrates, such as [https://en.wikipedia.org/wiki/Steroid steroids] and [https://en.wikipedia.org/wiki/Thyroid_hormones thyroid hormones]. <Ref name = Qi> Qi X. and Li W. (2022). Unlocking the secrets to human NTCP structure. The Innovation 3(5), 100294. https://doi.org/10.1016/j.xinn.2022.100294 </ref> Thus the channel provides specificity while preventing leakage of other substrates. When observing the relevant <scene name='95/952722/Bile_salts_res/1'>bile salt binding residues</scene> it is shown that some residues form Van der Waals interactions while others will form dipole-dipole or ionic interactions with bile salt substrates. The core domain appears to contribute most of the polar domains, while the panel domain contributes more hydrophobic residues. | The <scene name='95/952721/Amphipathic_patterns/2'>amphipathic pore</scene> is also characteristic of NTCP. The pore surface remains {{Template:ColorKey_Hydrophobic}}, while lining of the open pore state is largely {{Template:ColorKey_Polar}}. However, in the "inward-facing conformation" the polar pore residues are inaccessible. When the pore is closed only the surface hydrophobic residues are observed. As the pore opens up inner polar residues become accessible allowing for the binding of substrates. <Ref name = The pattern of hydrophobic and polar residues within the pore is believed to follow similar amphipathic patterns within taurocholate and other NTCP substrates, such as [https://en.wikipedia.org/wiki/Steroid steroids] and [https://en.wikipedia.org/wiki/Thyroid_hormones thyroid hormones]. <Ref name = Qi> Qi X. and Li W. (2022). Unlocking the secrets to human NTCP structure. The Innovation 3(5), 100294. https://doi.org/10.1016/j.xinn.2022.100294 </ref> Thus the channel provides specificity while preventing leakage of other substrates. When observing the relevant <scene name='95/952722/Bile_salts_res/1'>bile salt binding residues</scene> it is shown that some residues form Van der Waals interactions while others will form dipole-dipole or ionic interactions with bile salt substrates. The core domain appears to contribute most of the polar domains, while the panel domain contributes more hydrophobic residues. | ||
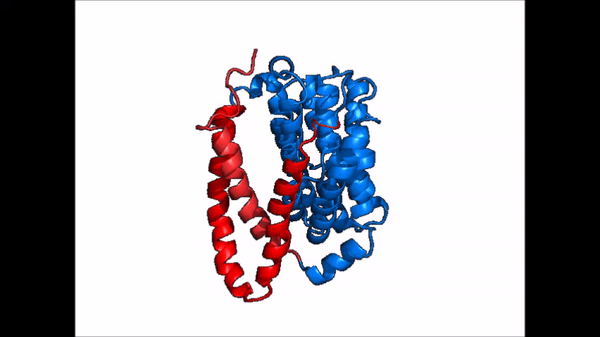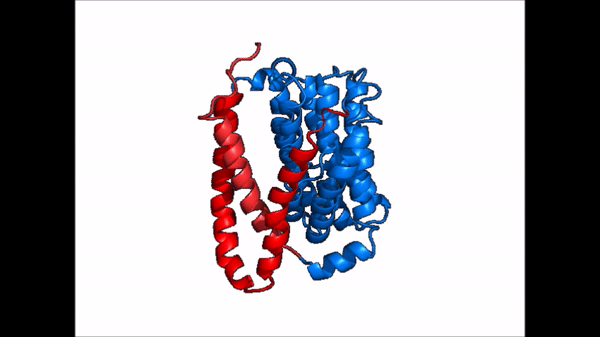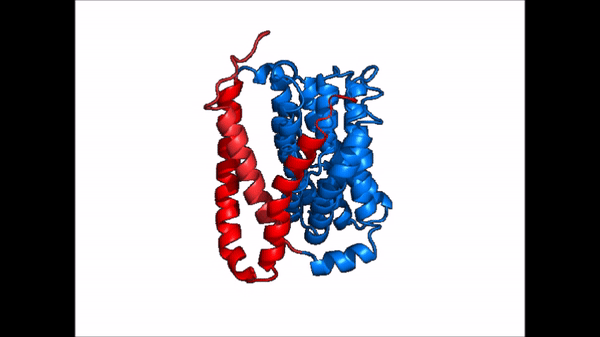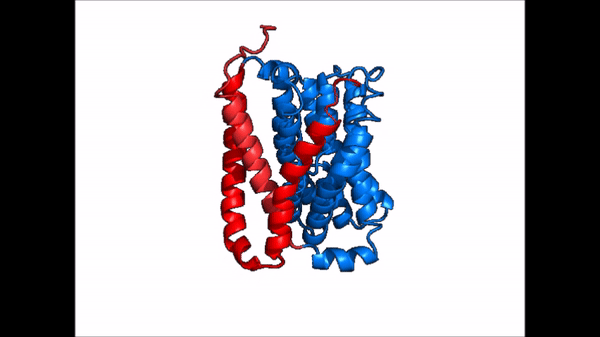
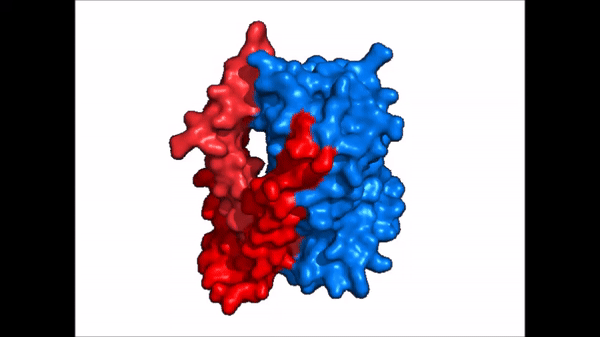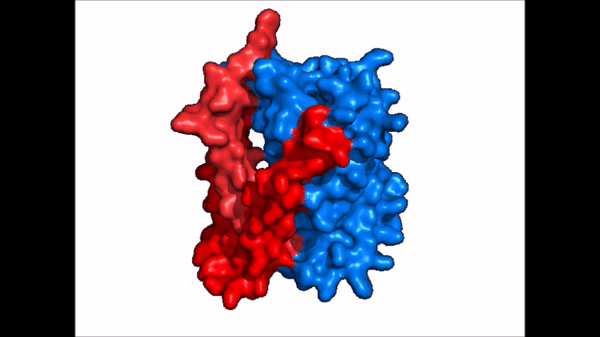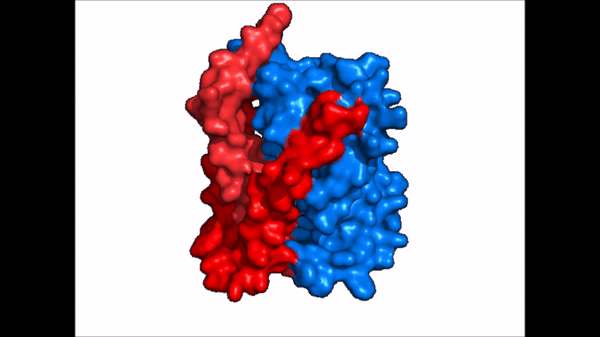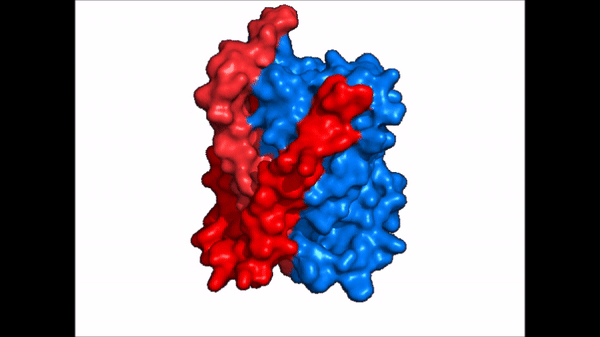
| + | == Conformational Change == | ||
| + | <table align='right' border='0' width='0' cellpadding='0' bgcolor='#d0d0d0' hspace='0'><tr><td rowspan='2'> </td><td bgcolor='#e8e8e8'>[[Image:Cartoon_NTCP_confchange.gif|100px]]</td></tr><tr><td bgcolor='#e8e8e8'>Cartoon representation of NTCP conformational change.</td></tr></table> | ||
| + | |||
| + | <table align='right' border='0' width='4' cellpadding='0' bgcolor='#d0d0d0' hspace='0'><tr><td rowspan='2'> </td><td bgcolor='#e8e8e8'>[[Image:Surface_NTCP_confchange.gif]]</td></tr><tr><td bgcolor='#e8e8e8'>Cartoon representation of NTCP conformational change.</td></tr></table> | ||
== Bile Salt Transport == | == Bile Salt Transport == | ||
Revision as of 15:55, 7 April 2023
Sodium Bile Salt Co-Transporting Protein
| |||||||||||
References
- ↑ Stieger B. The role of the sodium-taurocholate cotransporting polypeptide (NTCP) and of the bile salt export pump (BSEP) in physiology and pathophysiology of bile formation. Handb Exp Pharmacol. 2011;(201):205-59. doi: 10.1007/978-3-642-14541-4_5. PMID: 21103971. DOI: DOI: 10.1007/978-3-642-14541-4_5.
- ↑ Geyer, J., Wilke, T. & Petzinger, E. The solute carrier family SLC10: more than a family of bile acid transporters regarding function and phylogenetic relationships. Naunyn Schmied Arch Pharmacol 372, 413–431 (2006). https://doi.org/10.1007/s00210-006-0043-8
- ↑ 3.0 3.1 3.2 Park, JH., Iwamoto, M., Yun, JH. et al. Structural insights into the HBV receptor and bile acid transporter NTCP. Nature 606, 1027–1031 (2022). https://doi.org/10.1038/s41586-022-04857-0.
- ↑ 4.0 4.1 Goutam, K., Ielasi, F.S., Pardon, E. et al. Structural basis of sodium-dependent bile salt uptake into the liver. Nature 606, 1015–1020 (2022). DOI: 10.1038/s41586-022-04723-z.
- ↑ Qi X. and Li W. (2022). Unlocking the secrets to human NTCP structure. The Innovation 3(5), 100294. https://doi.org/10.1016/j.xinn.2022.100294
- ↑ 6.0 6.1 6.2 Liu, H., Irobalieva, R.N., Bang-Sørensen, R. et al. Structure of human NTCP reveals the basis of recognition and sodium-driven transport of bile salts into the liver. Cell Res 32, 773–776 (2022). https://doi.org/10.1038/s41422-022-00680-4
- ↑ 7.0 7.1 7.2 7.3 Asami, J., Kimura, K.T., Fujita-Fujiharu, Y. et al. Structure of the bile acid transporter and HBV receptor NTCP. Nature 606, 1021–1026 (2022). https://doi.org/10.1038/s41586-022-04845-4
- ↑ 8.0 8.1 Herrscher C, Roingeard P, Blanchard E. Hepatitis B Virus Entry into Cells. Cells. 2020 Jun 18;9(6):1486. doi: 10.3390/cells9061486. PMID: 32570893; PMCID: PMC7349259.