This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1790
From Proteopedia
(Difference between revisions)
| Line 12: | Line 12: | ||
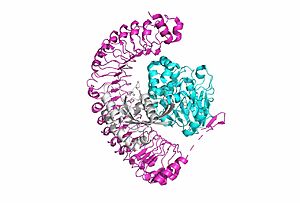
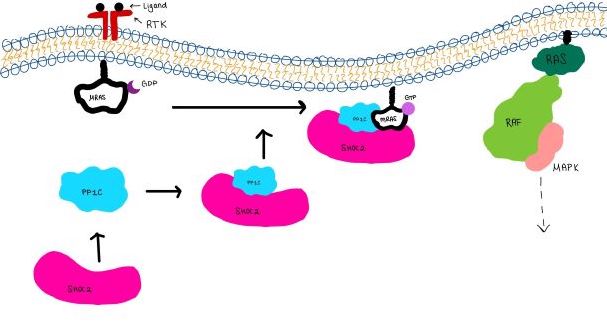
<scene name='95/952717/Shoc2/1'>SHOC2</scene> is a scaffold protein that is composed of 20 leucine-rich repeat domains that form a solenoid structure. The leucine rich region forms a concave hydrophobic core which is necessary for binding with PP1C and MRAS. | <scene name='95/952717/Shoc2/1'>SHOC2</scene> is a scaffold protein that is composed of 20 leucine-rich repeat domains that form a solenoid structure. The leucine rich region forms a concave hydrophobic core which is necessary for binding with PP1C and MRAS. | ||
==PP1C== | ==PP1C== | ||
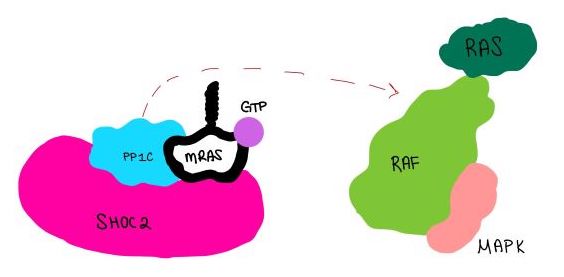
| - | <scene name='95/952717/Pp1c/1'>PP1C</scene> is a catalytic protein. After forming a ternary complex, the <scene name='95/952717/Pp1c_hydrophobic_patch/1'>hydrophobic active site</scene> on the protein interacts with Raf to act as a phosphatase and dephosphorylate Ser 259<ref | + | <scene name='95/952717/Pp1c/1'>PP1C</scene> is a catalytic protein. After forming a ternary complex, the <scene name='95/952717/Pp1c_hydrophobic_patch/1'>hydrophobic active site</scene> on the protein interacts with Raf to act as a phosphatase and dephosphorylate Ser 259<ref>PMID:35830882</ref>.. |
==MRAS== | ==MRAS== | ||
| - | <scene name='95/952717/Mras/2'>MRAS</scene> is a membrane bound structure that aids the complex in localizing near other structures such as the RAS-RAF-MAPK complex in order to initiate downstream signaling. In its inactive state, MRAS is bound to GDP. When signaled by [https://www.ncbi.nlm.nih.gov/books/NBK442024/. growth factors], the GDP is exchanged for GTP. The now <scene name='95/952718/Zoom_in_gtp/1'>GTP bound MRAS</scene> undergoes a conformational change of the <scene name='95/952716/Ras-switch-zoomed/1'>switch I and switch II regions</scene>. This conformational change activates the protein allowing it to bind more easily with the SHOC2-PP1C complex. In comparison to other RAS proteins, MRAS has a greater affinity for the SHOC2-PP1C complex<ref | + | <scene name='95/952717/Mras/2'>MRAS</scene> is a membrane bound structure that aids the complex in localizing near other structures such as the RAS-RAF-MAPK complex in order to initiate downstream signaling. In its inactive state, MRAS is bound to GDP. When signaled by [https://www.ncbi.nlm.nih.gov/books/NBK442024/. growth factors], the GDP is exchanged for GTP. The now <scene name='95/952718/Zoom_in_gtp/1'>GTP bound MRAS</scene> undergoes a conformational change of the <scene name='95/952716/Ras-switch-zoomed/1'>switch I and switch II regions</scene>. This conformational change activates the protein allowing it to bind more easily with the SHOC2-PP1C complex. In comparison to other RAS proteins, MRAS has a greater affinity for the SHOC2-PP1C complex<ref>PMID:35970881</ref>. |
| Line 39: | Line 39: | ||
==RASopathies== | ==RASopathies== | ||
| - | RASopathy is a broad term used to describe developmental syndromes that stem from [https://www.sciencedirect.com/topics/medicine-and-dentistry/germline-mutation. germline mutations] of proteins along the RAS/MAPK pathway such as SHOC2, PP1C, and MRAS. These mutations can be either gain or loss of function. Rasopathies can also lead to cancer<ref | + | RASopathy is a broad term used to describe developmental syndromes that stem from [https://www.sciencedirect.com/topics/medicine-and-dentistry/germline-mutation. germline mutations] of proteins along the RAS/MAPK pathway such as SHOC2, PP1C, and MRAS. These mutations can be either gain or loss of function. Rasopathies can also lead to cancer<ref>PMID:23875798</ref>. |
==Cancer== | ==Cancer== | ||
Revision as of 15:18, 7 April 2023
| |||||||||||