This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1790
From Proteopedia
(Difference between revisions)
| Line 14: | Line 14: | ||
[[Image:ActiveSiteProto.png|500 px|thumb|'''Figure 2:'''Active site of PP1C on SMP.</div></font>]] | [[Image:ActiveSiteProto.png|500 px|thumb|'''Figure 2:'''Active site of PP1C on SMP.</div></font>]] | ||
==MRAS== | ==MRAS== | ||
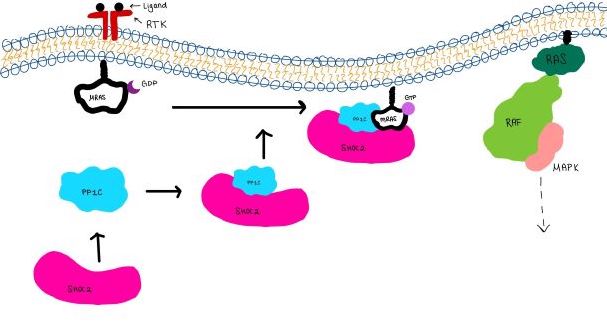
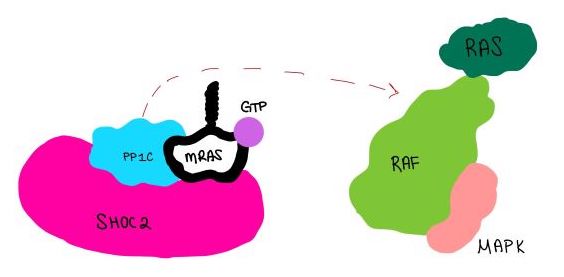
| - | <scene name='95/952717/Mras/2'>MRAS</scene> is a membrane-bound structure that aids the complex in localizing near other structures such as the RAS-RAF-MAPK complex in order to initiate downstream signaling. In its inactive state, MRAS is bound to GDP. When signaled by growth factors, the GDP is exchanged for GTP. The now <scene name='95/952718/Zoom_in_gtp/1'>GTP bound MRAS</scene> undergoes a conformational change of the <scene name='95/952716/Ras-switch-zoomed/1'>switch I and switch II regions</scene>. This conformational change activates the protein allowing it to bind with the SHOC2-PP1C complex. Without the conformational change when GDP is exchanged to GTP, the GDP-MRAS wouldn't be able to bind to SHOC2 because of steric clashing. In comparison to other RAS proteins, MRAS has a greater affinity for the SHOC2-PP1C complex<ref name=”Kubicek”> | + | <scene name='95/952717/Mras/2'>MRAS</scene> is a membrane-bound structure that aids the complex in localizing near other structures such as the RAS-RAF-MAPK complex in order to initiate downstream signaling. In its inactive state, MRAS is bound to GDP. When signaled by growth factors, the GDP is exchanged for GTP. The now <scene name='95/952718/Zoom_in_gtp/1'>GTP bound MRAS</scene> undergoes a conformational change of the <scene name='95/952716/Ras-switch-zoomed/1'>switch I and switch II regions</scene>. This conformational change activates the protein allowing it to bind with the SHOC2-PP1C complex. Without the conformational change when GDP is exchanged to GTP, the GDP-MRAS wouldn't be able to bind to SHOC2 because of steric clashing. In comparison to other RAS proteins, MRAS has a greater affinity for the SHOC2-PP1C complex<ref name=”Kubicek”>Kubicek M, Pacher M, Abraham D, Podar K, Eulitz M, Baccarini M. Dephosphorylation of Ser-259 regulates Raf-1 membrane association. J Biol Chem. 2002 Mar 8;277(10):7913-9. [http://10.1074/jbc.M108733200 doi: 10.1074/jbc.M108733200.]</ref>. MRAS engages the SHOC2-PP1C complex and RAF on the same surface indicating that for RAF signaling two separate active MRASs are needed. Having two MRASs also help with the co-localization of PP1C to the NTpS region on RAF. |
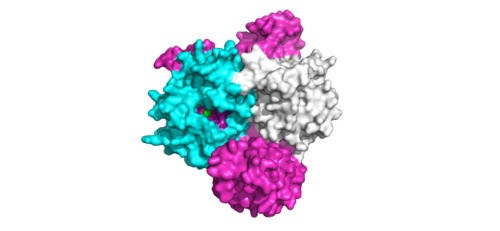
[[Image:Switches.png|500 px|thumb|'''Figure 2:'''Steric clashing of Switch I and II of GDP bound MRAS, in green, with the surface of SHOC2, in magenta. GTP-bound MRAS, in white, shows no steric clashing with SHOC2s surface.</div></font>]] | [[Image:Switches.png|500 px|thumb|'''Figure 2:'''Steric clashing of Switch I and II of GDP bound MRAS, in green, with the surface of SHOC2, in magenta. GTP-bound MRAS, in white, shows no steric clashing with SHOC2s surface.</div></font>]] | ||
| Line 25: | Line 25: | ||
==SHOC2 and MRAS== | ==SHOC2 and MRAS== | ||
| - | MRAS is initially bound to GDP causing it to be in its inactive state. This form cannot bind to the SHOC2-PP1C complex due to steric clashing. Once GDP is exchanged for GTP to activate the protein, <scene name='95/952716/conformational changes/1'>SHOC2-MRAS (residues)</scene> occur within the switch I and switch II regions to allow <scene name='95/952716/MRAS to interact with SHOC2/2'>SHOC2-MRAS(full-image)</scene>. These <scene name='95/952716/Scho2-mras-interactions/1'>interactions</scene> include hydrogen bonds and pi stacking. The primary hydrogen bonds are R288-Q71 and R177-E47. Pi staking occurs at R104-R83 <ref name=”Lavoie”>PMID:35970881</ref>. | + | MRAS is initially bound to GDP causing it to be in its inactive state. This form cannot bind to the SHOC2-PP1C complex due to steric clashing. Once GDP is exchanged for GTP to activate the protein, <scene name='95/952716/conformational changes/1'>SHOC2-MRAS (residues)</scene> occur within the switch I and switch II regions to allow <scene name='95/952716/MRAS to interact with SHOC2/2'>SHOC2-MRAS(full-image)</scene>. These <scene name='95/952716/Scho2-mras-interactions/1'>interactions</scene> include hydrogen bonds and pi stacking. The primary hydrogen bonds are R288-Q71 and R177-E47. Pi staking occurs at R104-R83 <ref name=”Lavoie”>Lavoie H, Therrien M. Structural keys unlock RAS-MAPK cellular signalling pathway. Nature. 2022 Sep;609(7926):248-249. [http://dx.doi.org/10.1038/d41586-022-02189-7 doi: 10.1038/d41586-022-02189-7. PMID: 35970881.]</ref>. |
==PP1C and MRAS== | ==PP1C and MRAS== | ||
Revision as of 16:44, 7 April 2023
| |||||||||||