Introduction
(SMP) is a ternary holophosphotase complex formed by the individual proteins: SHOC2, PP1C, and MRAS. The SMP complex is involved in signaling the initiation of MAPK pathways, which is responsible for cellular growth and development, cell proliferation, and apoptosis [1]. Formation of this complex begins with an extracellular signal binding to a membrane embedded receptor tyrosine kinase receptor(RTK) [1]. This causes membrane-bound MRAS to exchange GDP for GTP. Initiating the SMP complex formation at the plasma membrane consists of the SHOC2 and PP1C binding first. When the MRAS exchanges GDP to GTP, it then assembles with the combined SHOC2 and PP1C. Based on MRAS targeting, PP1C catalyzes the dephosphorylation of the N-terminal phosphoserine (NTpS) on the RAF complex leading to the amplification of MAPK signaling [1]. In a normal cell, this would regulate cell proliferation but dysfunction in the ternary complex has shown signs to lead to tumor formation due to unregulated cell growth [1].

Figure 1: Mechanism for Shoc2-MRAS-PP1C

Figure 2: PP1C phosphorylates Serine 259.
Overall Structure
SHOC2
is a scaffold protein composed of 20 leucine-rich repeat (LRR) domains that form a solenoid structure [1]. The leucine rich region forms a concave hydrophobic core which is necessary for binding with PP1C and MRAS. SHOC2 is the crucial mediator for SHOC2-PP1C-MRAS complex formation [1]. The leucine rich domain is very important in creating selectivity for the PP1C protein, as that protein is used for so many other complex pathways [1]. The LRR domains are stabilized by an N-terminal flanking 𝝰-helix and a C-terminal helix-turn-helix [2]. Alongside the conserved leucine residues in the LRR domain, there is a group of conserved asparagine residues that creates a stabilizing “asparagine ladder” that is necessary for the LRR fold, giving the SHOC2 its concave structure [2].
SHOC2 is also capable of causing various forms of Rasopathies. A common one is caused by a mutation known as p.S2G [3]. This mutation causes the formation of an additional 14-carbon saturated fatty acid chain on the N-terminal glycine of SHOC2 [3]. This causes SHOC2 to become attached to the cell membrane, resulting in a prolonged dephosphorylation of RAF by PP1C [3]. With this abnormality, there is overexpression of the MAPK pathway and increased cell proliferation genes [3]
PP1C
is a phosphatase. After forming a ternary complex, the hydrophobic active site on PP1C interacts with Raf and dephosphorylate Ser 259. PP1C's active site is adjacent to a hydrophobic binding pocket that binds to the C-terminal phosphoserine, located on the N-terminus of RAF, the target for dephosphorylation. PP1C can act as a phosphatase in the absence of SHOC2 but PP1C lacks intrinsic substrate selectively. The SMP complex formation endows PP1C with specificity for RAF [1].
The mechanism that PP1C uses to catalyze the dephosphorylation is mainly through donating a hydrogen atom to a phosphate group on the C-terminal of a phosphoserine on Raf [4]. This makes the phosphate group a good leaving group and it breaks off [4]. This catalysis is done by the serine-threonine alpha catalytic site on PP1C [4]. In this catalytic site, there are two Manganese ions and one calcium ion [4]. These metal ions are necessary in stablizing this catalytic site and there are a lot of polar negative residues in this region [4]
MRAS
is a monomeric GTPase. MRAS is membrane-bound due to post-translational lipidation which allows the protein to interact with the inner membrane leaflet. [5] MRAS localizes the SMP complex near RAF and other components of downstream signaling. The region of MRAS not directly bound to the membrane binds SHOC2 and PP1C to orient the complex such that PP1C’s active site faces the serine that will get dephosphorylated on RAF. MRAS also controls SMP complex formation in connection with extracellular signaling based on its dualistic switching between its inactive and active state. In its inactive state, MRAS is bound to GDP. When signaled by growth factors, the GDP is exchanged for GTP when a ligand binds to the RTK [1]. The now undergoes a conformational change of the . These regions are the major binding sites with SHOC2. This conformational change activates MRAS allowing it to bind with the SHOC2-PP1C complex. In its inactive GDP-bound state, MRAS is sterically occluded from binding SHOC2. For example, R83 of GDP-bound MRAS directly clashes with SHOC2 as shown in figure 2. In comparison to other RAS proteins such as H/K/NRAS, MRAS has a greater affinity for the SHOC2-PP1C complex[4]. This indicates that the specific structure of MRAS is necessary for SMP function. While MRAS engages the SHOC2-PP1C complex to bring the complex to the membrane, an additional membrane-bound RAS binds RAF nearby. This binding is also stimulated by ligand binding to the RTK. This indicates that for full RAF activation and continuous signaling of Raf, two separate active RAS proteins are needed. Having two MRASs also help with the co-localization of PP1C to the NTpS region on RAF. To inactivate Raf signaling, MRAS uses its intrinsic GTPase to remove the activating gamma-phosphate on GTP. In the GDP-bound state, switch I and II move to the position shown in green in Figure 2. This inactivates SHOC2 binding due to steric clashing which causes the SMP structure to dissociate.
Key Ligand Interactions
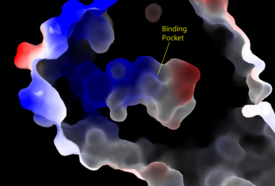
Figure 3: Electrostatic illustration of the amphipathic binding pocket of the LPA
1 receptor. This binding pocket was revealed by cutting away the exterior or the protein. This binding pocket, located in the interior of the protein, has both polar and nonpolar regions. The blue and red coloration highlight the positively and negatively charged regions, respectively, and the white color shows the nonpolar region of the binding pocket.
SHOC2 and PP1C
SHOC2 and MRAS
PP1C and MRAS
Signaling Pathway
Disease Relevance
Cancer
RASopathies
Future Studies
3D structures of lysophosphatidic acid receptor
4z34, 4z35, 4z36 - hLPA1 + antagonist - human
2lq4 – hLPA1 second extracellular loop – NMR
4p0c – hLPA2/NHERF2
5xsz – LPA6A (mutant) – zebra fish
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Hauseman ZJ, Fodor M, Dhembi A, Viscomi J, Egli D, Bleu M, Katz S, Park E, Jang DM, Porter KA, Meili F, Guo H, Kerr G, Molle S, Velez-Vega C, Beyer KS, Galli GG, Maira SM, Stams T, Clark K, Eck MJ, Tordella L, Thoma CR, King DA. Structure of the MRAS-SHOC2-PP1C phosphatase complex. Nature. 2022 Jul 13. pii: 10.1038/s41586-022-05086-1. doi:, 10.1038/s41586-022-05086-1. PMID:35830882 doi:http://dx.doi.org/10.1038/s41586-022-05086-1
- ↑ 2.0 2.1 Kwon JJ, Hajian B, Bian Y, Young LC, Amor AJ, Fuller JR, Fraley CV, Sykes AM, So J, Pan J, Baker L, Lee SJ, Wheeler DB, Mayhew DL, Persky NS, Yang X, Root DE, Barsotti AM, Stamford AW, Perry CK, Burgin A, McCormick F, Lemke CT, Hahn WC, Aguirre AJ. Structure-function analysis of the SHOC2-MRAS-PP1C holophosphatase complex. Nature. 2022 Jul 13. pii: 10.1038/s41586-022-04928-2. doi:, 10.1038/s41586-022-04928-2. PMID:35831509 doi:http://dx.doi.org/10.1038/s41586-022-04928-2
- ↑ 3.0 3.1 3.2 3.3 Rauen KA. The RASopathies. Annu Rev Genomics Hum Genet. 2013;14:355-69. doi: 10.1146/annurev-genom-091212-153523.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 Kubicek M, Pacher M, Abraham D, Podar K, Eulitz M, Baccarini M. Dephosphorylation of Ser-259 regulates Raf-1 membrane association. J Biol Chem. 2002 Mar 8;277(10):7913-9. doi: 10.1074/jbc.M108733200.
- ↑ Seabra MC. Membrane association and targeting of prenylated Ras-like GTPases. Cell Signal. 1998 Mar;10(3):167-72. PMID:9607139 doi:10.1016/s0898-6568(97)00120-4
Proteopedia Resources
Category:Lysophosphatidic acid binding
Category:Lysophosphatidic acid
Butler University Proteopedia Pages
See also:



