Introduction

Fig.1 Taruocholic acid a crystalline bile acid Shown here is the structure in a line representation of taruocholic acid with stereochemistry and important hydrogens.
Sodium Taurocholate Co-Transporting Polypeptide, or NTCP, is a membrane transporter protein found in the plasma membrane of hepatocytes. NTCP's primary function is the transportation of taurocholates, or bile salts, (Fig. 1) into the liver and out of the liver to the small intestine. [1] Bile salts play various physiological roles in metabolism and digestion, but their main function is the emulsification of lipid droplets into smaller fragments. This enables lipases to break down the droplets into their monomers, or triglycerides which are then able to be digested. NTCP is part of the solute carrier superfamily, SLC10. NTCP is the founding member of the SLC10 family, first discovered in rat hepatocytes in 1978. [2] NTCP has a key role in enterohepatic circulation or bile salt recycling, and its unique ability to transport other solutes gives it therapeutic potential for lowering cholesterol and treating liver disease. [3]
In addition to its physiological role in bile salt transport, NTCP also serves as a binding site for hepatitis B virus and hepatitis D virus. [4] Understanding the HBV and HDV binding mechanism to NTCP may aid in the development of new viral inhibitors.
Structure
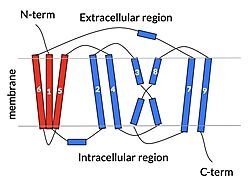
Fig. 2: cartoon depiction of NTCP topology. The panel domain is shown in red and the core domain is shown in blue. Each of the 9 transmembrane α helices are labeled and oriented according to their position embedded in the membrane.
Overview
The overall structure of NTCP is one continuous polypeptide chain containing embedded in the plasma membrane.[3] The N-terminus of this polypeptide chain extrudes into the extracellular region of the plasma membrane while the C-terminus juts into the intracellular region. NTCP contains : a core domain and a panel domain, which together channel opening and bile salt transport (Fig. 2). The (blue) contains 6 transmembrane α helices (TM2-4 and TM7-9) and demonstrates two-fold pseudosymmetry. The (red) consists of 3 transmembrane α helices (TM1 and TM5-6) and is asymmetrical. Within the core domain, a unique crossover between TM-3 and TM-8 creates an . The X motif contains the substrate binding site and essential residues for the conformational change required for transport. The core and panel domains are also connected by both extracellular and intracellular that are separate from the . All of these structural components of NTCP contribute to the transport of bile salts in and out of the liver.
Binding Sites
Sodium
NTCP, like other SLC10 family members, have . Many polar and negatively charged residues (68, 105, 106, 119, 123, 257, 261) form ion-dipole or dipole-dipole interactions with the sodium ions in these sites with a high level of conservation, suggesting sodium binding is coupled to bile salt transport. [3] Mutations in the X-motif near sodium binding sites also inhibit bile salt transport function, suggesting that sodium is required for salt binding.
[3] Sodium transport facilitates structural changes in NTCP from its typical open-pore state to an inward-facing (closed-pore) state. The inward-facing state is favored in the absence of sodium ions, while open-pore state is favored in the presence of sodium ions. [3] Gating of the channel with sodium in this way allows for sodium concentrations to regulate uptake of taurocholates. [3] When intracellular sodium levels are higher, open-pore state is favored allowing for the diffusion of taurocholates. However, when extracellular sodium levels are high, inward-facing state is favored preventing diffusion of taurocholates. [3] Overall, this suggests that thermodynamically favorable sodium transport is coupled to moving bile salts against their concentration gradient. [5]
Bile Salts
A key feature of NTCP is its which allows for bile salt transport across the hydrophilic membrane. The pore surface remains Hydrophobic, while the is largely Polar. In the inward-facing or closed-pore conformation, the polar pore residues are inaccessible. Only the surface hydrophobic residues are exposed. As the pore opens up, inner polar residues become accessible allowing for the binding of hydrophilic bile salts. The pattern of hydrophobic and polar residues within the pore matches the amphipathic patterns within taurocholates, steroids, and thyroid hormones. [6] Using this amphipathic pore, provides the channel with specificity while preventing leakage of other substrates. Essential form Van der Waals interactions with bile salt substrates, while others form dipole-dipole or ionic interactions. The core domain contributes most of the polar domains, while the panel domain contributes mainly hydrophobic surface.
Conformational Change
| 
|
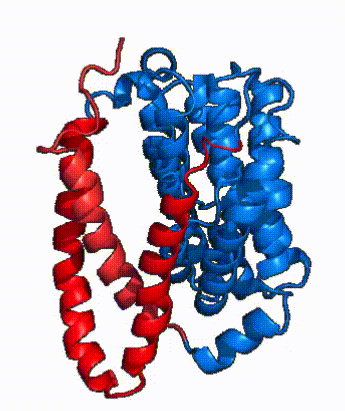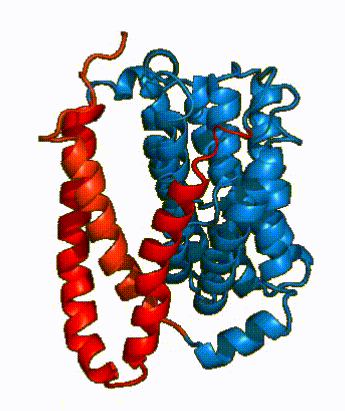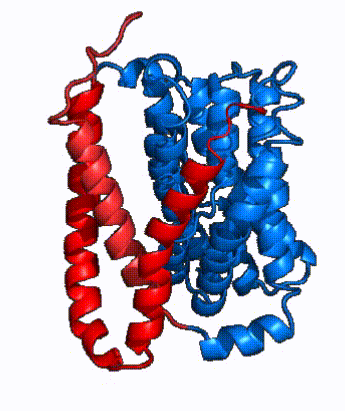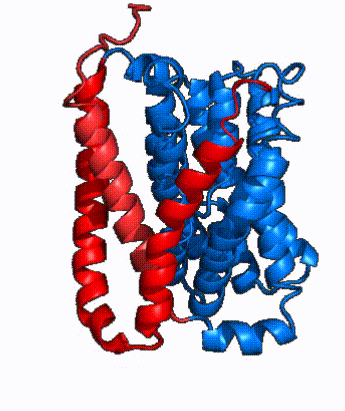
| Fig. 3: NTCP shown as cartoons with panel and core domains colored. NTCP is shown entirely as cartoons colored by domain. The panel domain helices can be seen moving to close the pore. Here NTCP is rotated 180° from how it is normally oriented and is alternating from open-pore to inward-facing conformation (7PQQ to 7PQG)
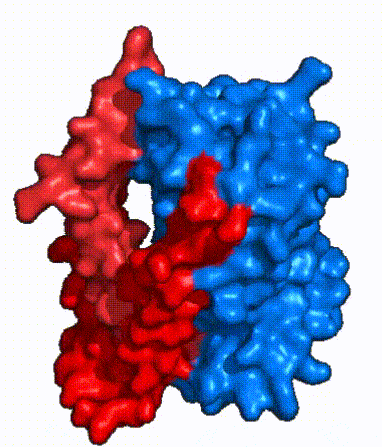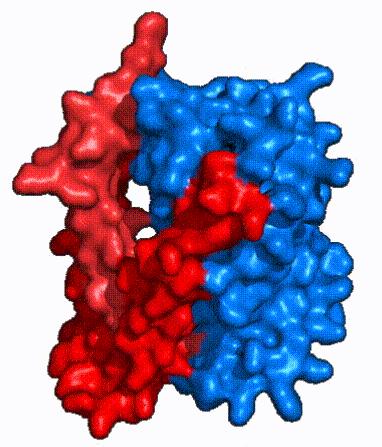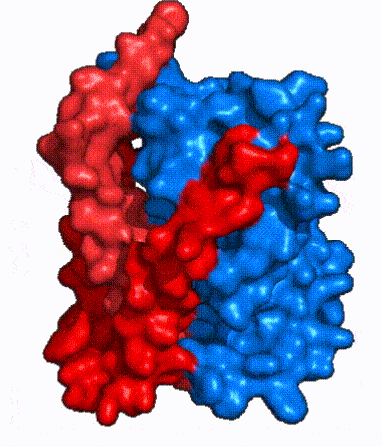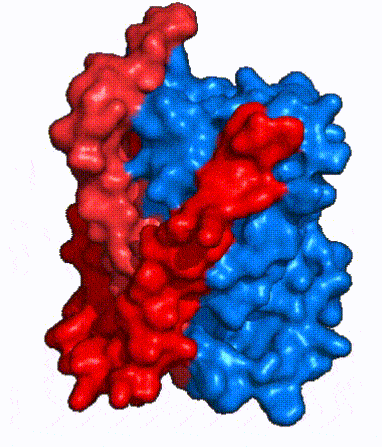
| Fig. 4: NTCP surface representation with panel and core domains colored. NTCP is shown as surface representation colored by domain. The panel domain can be seen shifting to close the pore. Here NTCP is rotated 180° from how it is normally oriented and is alternating from open-pore to inward-facing conformation (7PQQ to 7PQG)
|
In order to reveal these binding sites to initiate bile salt transport, NTCP exists in two different conformations; the and the . [3] NTCP undergoes a conformational change from inward facing to open pore which exposes the binding sites to the extracellular region to allow the sodium ions and bile salts to bind. NTCP utilizes an elevator-alternating mechanism [7] where one domain (core) does most of the translocation, and the other domain (panel) remains stationary. [8] In this movement, the and the rotate 20° with the panel domain moving 5 Å away from the core domain, which remains relatively rigid. This conformational change reveals the two sodium ion binding sites as well as the in the membrane. The movement of the panel domain is facilitated by located in the between the panel and core domains. (yellow) act as hinges that assist in the movement of the panel domain away from the core domain. [3]
Bile Salt Transport
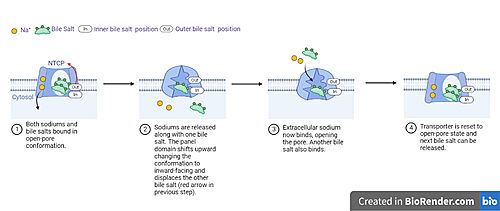
Fig. 5 Proposed process of NTCP bile salt transport. NTCP structure is shown in light blue with a white channel going through the protein. Sodium ions as yellow spheres and bile salts as green blobs with inner and outer binding sites labeled. Captions below each image detail how movement leads to transport of bile salts and conformational changes in NTCP throughout the different steps of the process.
A proposed pathway for NTCP bile salt transport starting and ending with open-pore states hypothesizes that both sodium ions are translocated with the transport of one bile salt.[5]. Only one taurocholate is transported at a time due to NTCP's two bile salt binding sites. An
that is closer to the cytoplasmic side of the membrane and an that is closer to the extracellular side of the membrane [5] In the open-pore state both Next both sodium ions are released into the cytoplasm along with the inner bile salt into the cytoplasm (Fig. 5). Once the sodium ions are released this will trigger the conformational change in NTCP. The in the pore, likely helping to prevent leakage. [5] Then the into the inner bile salt binding site by the movement from conformational change to the inward-facing, pore inaccessible state due to sodium ion release (Fig. 5). [5] Two new sodium ions then bind to NTCP from the extracellular side, favoring the open-pore state and also allowing for the binding of another outer bile salt (Fig 5). The and the process can then start again releasing the next inner bile salt with the translocation of the sodium ions into the cytoplasm. This mechanism melds typical patterns of gated channels and pumps in a new light as sodium ions control conformation and thus binding, displacement, and release of bile salts. [3]
HBV Binding and Infection
NTCP is the only entry receptor [9] into the liver for HBV and HDV. [8]These viruses are known to use (residues 84-87 and 157-165) on NTCP for binding and entry. The myristolated PreS1 domain of HBV binds to NTCP through the on NTCP containing residues 157-165 on the open pore surface. [8] These residues form part of the bile salt transport tunnel resulting in HBV binding and bile salt transport directly competing and interfering with one another. [8] The consisting of residues 84-87 found on the N-terminus of NTCP does not overlap with bile salt binding and may be used for the development of antivirals that do not inhibit bile uptake [4]. Other minor variations within NTCP provide species specificity for HBV or virus resistance, such as mutant S267F found in East Asia. [4] This S267F mutation is a single-nucleotide polymorphism, where a change in one nucleotide in the sequence has caused a lack of bile salt transport activity or viral infection. [4] It is hypothesized that due to the lack of bile salt transport in this mutation that the open-pore state during bile salt transport is necessary for HBV and HDV infection, suggesting the two functionally overlap. [4]
The exact mechanism by which NTCP mediates viral internalization is still being determined; however, current evidence suggests it works through endocytosis. [10] Once HBV is bound, the NTCP/HBV complex is taken into the cell where viral contents are dumped into the cytoplasm to then begin viral replication. HBV may also interact with other receptors or host cell factors, as cells overexpressing NTCP alone had low infection efficiency. [10]





