This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1794
From Proteopedia
(Difference between revisions)
| Line 37: | Line 37: | ||
|} | |} | ||
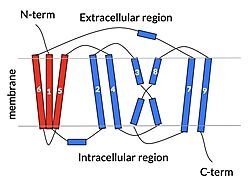
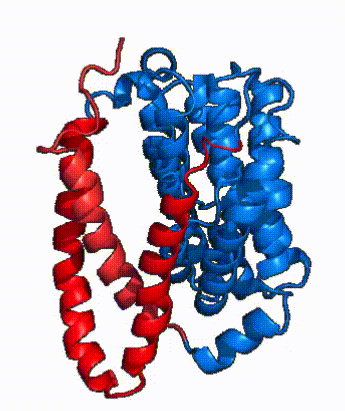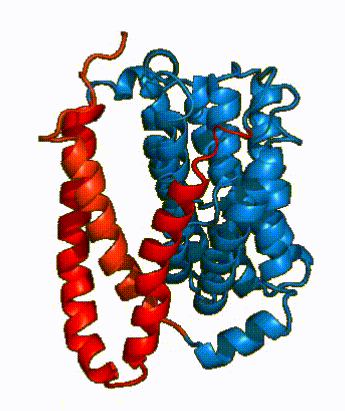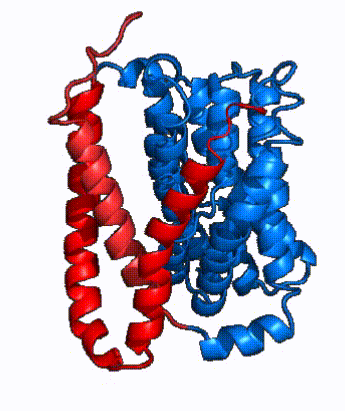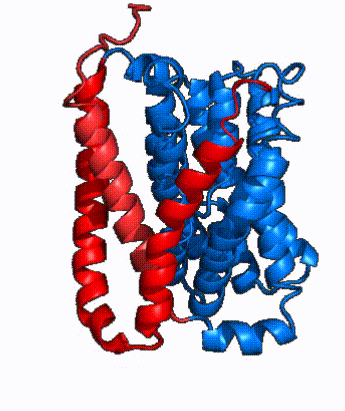
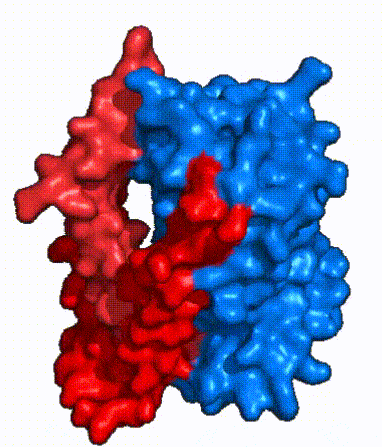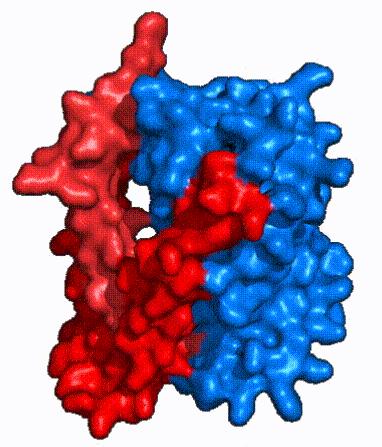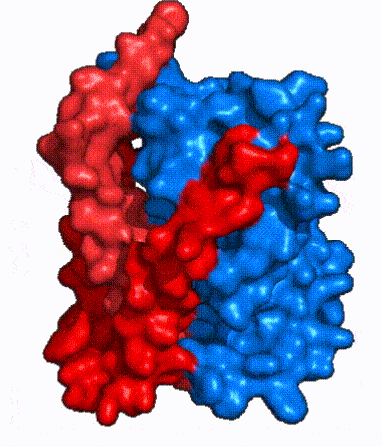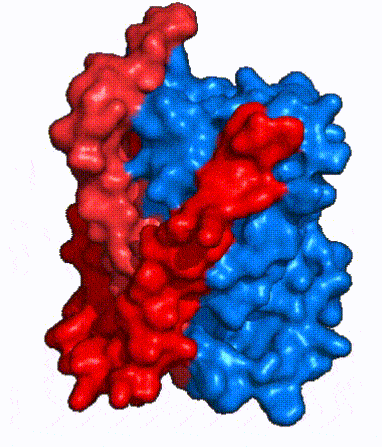
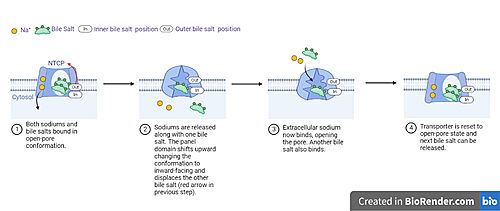
| - | In order to reveal these binding sites to initiate bile salt transport, NTCP exists in two different conformations; the <scene name='95/952722/Open_pore_conf/4'>open pore conformation</scene> and the <scene name='95/952722/Inward_facing_conf/1'>inward facing conformation</scene>. <ref name="Goutam"/> NTCP undergoes a conformational change from inward facing to open pore which exposes the binding sites to the extracellular region to allow the sodium ions and bile salts to bind. NTCP utilizes an [https://www.sciencedirect.com/science/article/pii/S0092867417302891 elevator-alternating mechanism] <Ref name = "Latorraca"> Latorraca, N. R.; Fastman, N. M.; Venkatakrishnan, A. J.; Frommer, W. B.; Dror, R. O.; Feng, L. Mechanism of Substrate Translocation in an Alternating Access Transporter. Cell 2017, 169 (1), 96–107. </ref> where one domain <font color=' | + | In order to reveal these binding sites to initiate bile salt transport, NTCP exists in two different conformations; the <scene name='95/952722/Open_pore_conf/4'>open pore conformation</scene> and the <scene name='95/952722/Inward_facing_conf/1'>inward facing conformation</scene>. <ref name="Goutam"/> NTCP undergoes a conformational change from inward facing to open pore which exposes the binding sites to the extracellular region to allow the sodium ions and bile salts to bind. NTCP utilizes an [https://www.sciencedirect.com/science/article/pii/S0092867417302891 elevator-alternating mechanism] <Ref name = "Latorraca"> Latorraca, N. R.; Fastman, N. M.; Venkatakrishnan, A. J.; Frommer, W. B.; Dror, R. O.; Feng, L. Mechanism of Substrate Translocation in an Alternating Access Transporter. Cell 2017, 169 (1), 96–107. </ref> where one domain <font color='red'><b>(panel)</b></font> does most of the translocation, and the other domain <font color='#6060ff'><b>(core)</b></font> remains stationary. <Ref name = "Asami"> Asami, J., Kimura, K.T., Fujita-Fujiharu, Y. et al.Structure of the bile acid transporter and HBV receptor NTCP. Nature 606, 1021–1026 (2022). https://doi.org/10.1038/s41586-022-04845-4 </ref> In this movement, the <scene name='95/952722/Ntcp_core_domain-_blue/8'>core domain</scene> and the <scene name='95/952722/Ntcp_panel_domain-_red/4'>panel domain</scene> rotate 20° with the <font color='red'><b>panel domain</b></font> moving 5 Å away from the <font color='#6060ff'><b>core domain</b></font>, which remains relatively rigid. This conformational change reveals the two sodium ion binding sites as well as the <scene name='95/952721/Amphipathic_patterns/2'>amphipathic pore</scene> in the membrane. The movement of the panel domain is facilitated by <scene name='95/952722/Pro_and_gly_hinges/5'>proline and glycine residues</scene> located in the <scene name='95/952722/Connector_helices/6'>connector helices</scene> between the panel and core domains. <scene name='95/952722/Pro_and_gly_hinges/7'>These residues</scene> <font color='#FCE205'><b>(yellow)</b></font> act as hinges that assist in the movement of the panel domain away from the core domain. <ref name="Goutam"/> |
== Bile Salt Transport == | == Bile Salt Transport == | ||
Revision as of 22:02, 20 April 2023
Contents |
Sodium Taurocholate Co-Transporting Polypeptide
| |||||||||||
References
- ↑ Stieger B. The role of the sodium-taurocholate cotransporting polypeptide (NTCP) and of the bile salt export pump (BSEP) in physiology and pathophysiology of bile formation. Handb Exp Pharmacol. 2011;(201):205-59. doi: 10.1007/978-3-642-14541-4_5. PMID: 21103971. DOI: DOI: 10.1007/978-3-642-14541-4_5.
- ↑ Geyer, J., Wilke, T. & Petzinger, E. The solute carrier family SLC10: more than a family of bile acid transporters regarding function and phylogenetic relationships. Naunyn Schmied Arch Pharmacol 372, 413–431 (2006). https://doi.org/10.1007/s00210-006-0043-8
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 3.9 Goutam, K., Ielasi, F.S., Pardon, E. et al. Structural basis of sodium-dependent bile salt uptake into the liver. Nature 606, 1015–1020 (2022). DOI: 10.1038/s41586-022-04723-z.
- ↑ 4.0 4.1 4.2 4.3 4.4 Park, JH., Iwamoto, M., Yun, JH. et al. Structural insights into the HBV receptor and bile acid transporter NTCP. Nature 606, 1027–1031 (2022). https://doi.org/10.1038/s41586-022-04857-0.
- ↑ 5.0 5.1 5.2 5.3 5.4 Liu, H., Irobalieva, R.N., Bang-Sørensen, R. et al. Structure of human NTCP reveals the basis of recognition and sodium-driven transport of bile salts into the liver. Cell Res 32, 773–776 (2022). https://doi.org/10.1038/s41422-022-00680-4
- ↑ Qi X. and Li W. (2022). Unlocking the secrets to human NTCP structure. The Innovation 3(5), 100294. https://doi.org/10.1016/j.xinn.2022.100294
- ↑ Latorraca, N. R.; Fastman, N. M.; Venkatakrishnan, A. J.; Frommer, W. B.; Dror, R. O.; Feng, L. Mechanism of Substrate Translocation in an Alternating Access Transporter. Cell 2017, 169 (1), 96–107.
- ↑ 8.0 8.1 8.2 8.3 Asami, J., Kimura, K.T., Fujita-Fujiharu, Y. et al.Structure of the bile acid transporter and HBV receptor NTCP. Nature 606, 1021–1026 (2022). https://doi.org/10.1038/s41586-022-04845-4
- ↑ Grove, J.; Marsh, M. The Cell Biology of Receptor-Mediated Virus Entry. Journal of Cell Biology 2011, 195 (7), 1071–1082.
- ↑ 10.0 10.1 Herrscher C, Roingeard P, Blanchard E. Hepatitis B Virus Entry into Cells. Cells. 2020 Jun 18;9(6):1486. doi: 10.3390/cells9061486. PMID: 32570893; PMCID: PMC7349259.
PDB Files
Bile salts and sodium ions bound: 7zyi Open-pore NTCP: 7pqq Inward-facing NTCP: 7pqg
Student Contributors
- Isabelle White
- Lena Barko