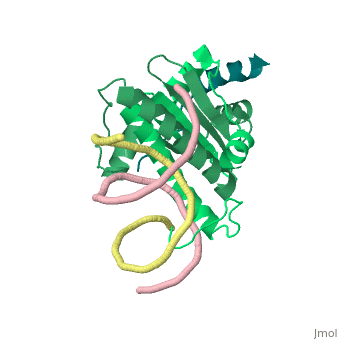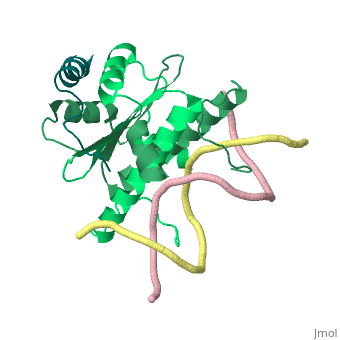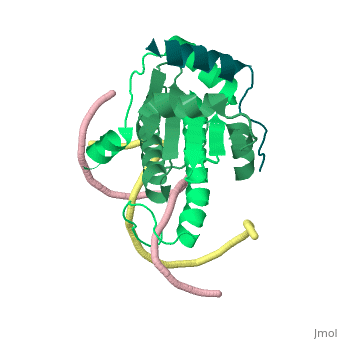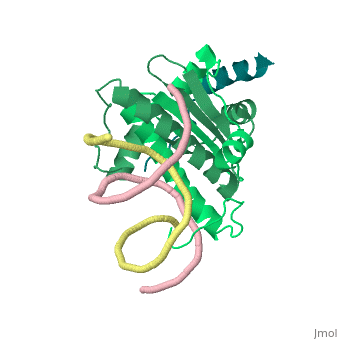This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1tqe
From Proteopedia
(New page: 200px<br /> <applet load="1tqe" size="450" color="white" frame="true" align="right" spinBox="true" caption="1tqe, resolution 2.7Å" /> '''Mechanism of recruit...) |
|||
| Line 1: | Line 1: | ||
| - | [[Image:1tqe.gif|left|200px]]<br /> | + | [[Image:1tqe.gif|left|200px]]<br /><applet load="1tqe" size="350" color="white" frame="true" align="right" spinBox="true" |
| - | <applet load="1tqe" size=" | + | |
caption="1tqe, resolution 2.7Å" /> | caption="1tqe, resolution 2.7Å" /> | ||
'''Mechanism of recruitment of class II histone deacetylases by myocyte enhancer factor-2'''<br /> | '''Mechanism of recruitment of class II histone deacetylases by myocyte enhancer factor-2'''<br /> | ||
==Overview== | ==Overview== | ||
| - | Class II histone deacetylases (HDACs) bind myocyte enhancer factor-2 | + | Class II histone deacetylases (HDACs) bind myocyte enhancer factor-2 (MEF2) and repress specific gene expression in a calcium-dependent manner. Despite their significant physiological functions in muscle, immune and neuronal cells, the mechanism of recruitment of class II HDACs by MEF2 is not well understood. Here, we have characterized the complex between the MEF2-binding motif of class II HDACs and the MADS-box/MEF2S domain of MEF2B by structural and biochemical methods. The crystal structure of a HDAC9/MEF2/DNA complex reveals that HDAC9 binds to a hydrophobic groove of the MEF2 dimer. The overall binding mode is similar to that seen in the Cabin1/MEF2/DNA complex. The detailed binding interactions at the HDAC9/MEF2 interface, however, show marked differences from those at the Cabin1/MEF2 interface. Our studies suggest a general mechanism by which class II HDACs and possibly other transcriptional co-regulators are recruited by MEF2. On the other hand, the differential binding between MEF2 and its various partners may confer specific regulatory and functional properties to MEF2 in distinct cellular processes. Such specificity provides a basis for selectively disrupting a particular MEF2/co-regulator complex by mutations or small molecules. |
==About this Structure== | ==About this Structure== | ||
| - | 1TQE is a [http://en.wikipedia.org/wiki/Protein_complex Protein complex] structure of sequences from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens] and [http://en.wikipedia.org/wiki/Mus_musculus Mus musculus]. Full crystallographic information is available from [http:// | + | 1TQE is a [http://en.wikipedia.org/wiki/Protein_complex Protein complex] structure of sequences from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens] and [http://en.wikipedia.org/wiki/Mus_musculus Mus musculus]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1TQE OCA]. |
==Reference== | ==Reference== | ||
| Line 18: | Line 17: | ||
[[Category: Han, A.]] | [[Category: Han, A.]] | ||
[[Category: He, J.]] | [[Category: He, J.]] | ||
| - | [[Category: Liu, J | + | [[Category: Liu, J O.]] |
[[Category: Wu, Y.]] | [[Category: Wu, Y.]] | ||
[[Category: co-repressor]] | [[Category: co-repressor]] | ||
| Line 25: | Line 24: | ||
[[Category: transcription]] | [[Category: transcription]] | ||
| - | ''Page seeded by [http:// | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Feb 21 15:16:17 2008'' |
Revision as of 13:16, 21 February 2008
|
Mechanism of recruitment of class II histone deacetylases by myocyte enhancer factor-2
Overview
Class II histone deacetylases (HDACs) bind myocyte enhancer factor-2 (MEF2) and repress specific gene expression in a calcium-dependent manner. Despite their significant physiological functions in muscle, immune and neuronal cells, the mechanism of recruitment of class II HDACs by MEF2 is not well understood. Here, we have characterized the complex between the MEF2-binding motif of class II HDACs and the MADS-box/MEF2S domain of MEF2B by structural and biochemical methods. The crystal structure of a HDAC9/MEF2/DNA complex reveals that HDAC9 binds to a hydrophobic groove of the MEF2 dimer. The overall binding mode is similar to that seen in the Cabin1/MEF2/DNA complex. The detailed binding interactions at the HDAC9/MEF2 interface, however, show marked differences from those at the Cabin1/MEF2 interface. Our studies suggest a general mechanism by which class II HDACs and possibly other transcriptional co-regulators are recruited by MEF2. On the other hand, the differential binding between MEF2 and its various partners may confer specific regulatory and functional properties to MEF2 in distinct cellular processes. Such specificity provides a basis for selectively disrupting a particular MEF2/co-regulator complex by mutations or small molecules.
About this Structure
1TQE is a Protein complex structure of sequences from Homo sapiens and Mus musculus. Full crystallographic information is available from OCA.
Reference
Mechanism of recruitment of class II histone deacetylases by myocyte enhancer factor-2., Han A, He J, Wu Y, Liu JO, Chen L, J Mol Biol. 2005 Jan 7;345(1):91-102. PMID:15567413
Page seeded by OCA on Thu Feb 21 15:16:17 2008
Categories: Homo sapiens | Mus musculus | Protein complex | Chen, L. | Han, A. | He, J. | Liu, J O. | Wu, Y. | Co-repressor | Hdac | Mef2 | Transcription