This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Jaelin Lunato/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 11: | Line 11: | ||
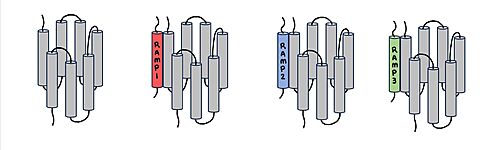
[[Image:RAMP_diagram.jpg|500px|right|thumb|'''Figure 1.''' Three different RAMPs to compose AMY1R, AMY2R, and AMY3R when associated with the calcitonin receptor (shown in grey). RAMP1 is in red, RAMP2 is in blue, and RAMP3 is in green.]] AMYR has three domains: extracellular, transmembrane, and intracellular. The CTR and the RAMP have both <scene name='10/1037495/Extracellular_domain/7'>extracellular domains</scene> and <scene name='10/1037495/Transmembrane/6'>transmembrane domains</scene>. The calcitonin receptor is a 7-pass helix chain to which RAMP binds to become the amylin receptor. The ligand, amylin, binds within the transmembrane domain. The [https://en.wikipedia.org/wiki/G_protein G protein] is located <scene name='10/1037495/Intracellular_domain/2'>inside the cell</scene>. The location of each component of the amylin receptor is essential in determining structure and function. For example, it is important for the G protein to be located inside the cell in order to initiate a signal cascade for cell signaling. | [[Image:RAMP_diagram.jpg|500px|right|thumb|'''Figure 1.''' Three different RAMPs to compose AMY1R, AMY2R, and AMY3R when associated with the calcitonin receptor (shown in grey). RAMP1 is in red, RAMP2 is in blue, and RAMP3 is in green.]] AMYR has three domains: extracellular, transmembrane, and intracellular. The CTR and the RAMP have both <scene name='10/1037495/Extracellular_domain/7'>extracellular domains</scene> and <scene name='10/1037495/Transmembrane/6'>transmembrane domains</scene>. The calcitonin receptor is a 7-pass helix chain to which RAMP binds to become the amylin receptor. The ligand, amylin, binds within the transmembrane domain. The [https://en.wikipedia.org/wiki/G_protein G protein] is located <scene name='10/1037495/Intracellular_domain/2'>inside the cell</scene>. The location of each component of the amylin receptor is essential in determining structure and function. For example, it is important for the G protein to be located inside the cell in order to initiate a signal cascade for cell signaling. | ||
| - | === | + | === CTR and RAMP Heterodimer === |
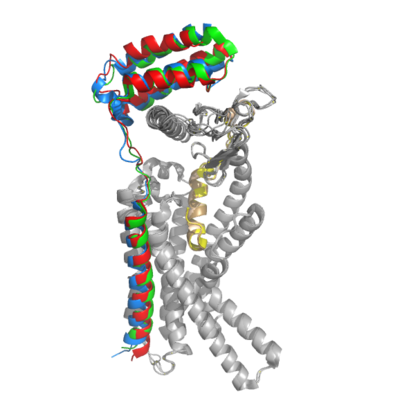
[[Image:Overlay of RAMPs.png|400 px|right|thumb|'''Figure 2.''' Superimposition of RAMP1, RAMP2, and RAMP3. RAMP1 is red, RAMP2 is blue, and RAMP3 is green. The amylin ligand is dark yellow, and the calcitonin ligand is pale yellow.]] AMYR is a heterodimer of a calcitonin receptor and a receptor activity-modifying protein. There are three different RAMPs, RAMP1, RAMP2, and RAMP3, that compose AMY1R, AMY2R, and AMY3R when associated with the CTR (Figure 1). The three different RAMPs are structurally similar to each other, so all three RAMPs are able to bind to the CTR without any modification of the CTR (Figure 2). | [[Image:Overlay of RAMPs.png|400 px|right|thumb|'''Figure 2.''' Superimposition of RAMP1, RAMP2, and RAMP3. RAMP1 is red, RAMP2 is blue, and RAMP3 is green. The amylin ligand is dark yellow, and the calcitonin ligand is pale yellow.]] AMYR is a heterodimer of a calcitonin receptor and a receptor activity-modifying protein. There are three different RAMPs, RAMP1, RAMP2, and RAMP3, that compose AMY1R, AMY2R, and AMY3R when associated with the CTR (Figure 1). The three different RAMPs are structurally similar to each other, so all three RAMPs are able to bind to the CTR without any modification of the CTR (Figure 2). | ||
In the absence of a RAMP, the calcitonin receptor has greater affinity for calcitonin than amylin, but because the <scene name='10/1037495/Overlay_of_ligands/1'>two ligands are structurally similar</scene>, both calcitonin and amylin can bind to the CTR without any modification of the receptor. The two ligands have many conserved residues (highlighted in blue), and this shows that the two ligands share many chemical properties which explains why they are structurally similar. In addition to having many conserved residues, both ligands also have an amidated C-terminus (Figure 3). When the CTR is bound to a RAMP, the complex becomes the AMYR and has greater affinity for the <scene name='10/1037495/Ligand_in_membrane/2'>amylin ligand</scene> relative to the calcitonin ligand. Therefore, the RAMP is essential to AMYR because it causes AMYR to have greater affinity for the amylin ligand rather than the calcitonin ligand. [[Image:Sequence alignment ligands.png|500px|left|thumb|'''Figure 3.''' Sequence alignment of rat amylin and salmon calcitonin. Conserved residues are highlighted in blue.]] | In the absence of a RAMP, the calcitonin receptor has greater affinity for calcitonin than amylin, but because the <scene name='10/1037495/Overlay_of_ligands/1'>two ligands are structurally similar</scene>, both calcitonin and amylin can bind to the CTR without any modification of the receptor. The two ligands have many conserved residues (highlighted in blue), and this shows that the two ligands share many chemical properties which explains why they are structurally similar. In addition to having many conserved residues, both ligands also have an amidated C-terminus (Figure 3). When the CTR is bound to a RAMP, the complex becomes the AMYR and has greater affinity for the <scene name='10/1037495/Ligand_in_membrane/2'>amylin ligand</scene> relative to the calcitonin ligand. Therefore, the RAMP is essential to AMYR because it causes AMYR to have greater affinity for the amylin ligand rather than the calcitonin ligand. [[Image:Sequence alignment ligands.png|500px|left|thumb|'''Figure 3.''' Sequence alignment of rat amylin and salmon calcitonin. Conserved residues are highlighted in blue.]] | ||
| - | + | == Ligands == | |
The two major ligands of the calcitonin receptor are [https://en.wikipedia.org/wiki/Calcitonin calcitonin] and [https://en.wikipedia.org/wiki/Amylin amylin]. Calcitonin is a peptide hormone secreted from the thyroid, and it is involved in the regulation of calcium and phosphate in the blood. Amylin is also a hormone and is secreted by pancreatic beta cells. Amylin binding activates many different biological processes affecting different systems with the human body. For example, amylin binding can affect the immune system, the central nervous system, and the satiation system in the brain (i.e., the area postrema). There are two required post-translational modifications of amylin in order for the ligand to have any bioactivity: (1) <scene name='10/1037495/C-term_amide/2'>amidation of the C-terminus</scene> and (2) a <scene name='10/1037495/Amylin_disulfide_bond2/4'>disulfide bond</scene> between C2 and C7. | The two major ligands of the calcitonin receptor are [https://en.wikipedia.org/wiki/Calcitonin calcitonin] and [https://en.wikipedia.org/wiki/Amylin amylin]. Calcitonin is a peptide hormone secreted from the thyroid, and it is involved in the regulation of calcium and phosphate in the blood. Amylin is also a hormone and is secreted by pancreatic beta cells. Amylin binding activates many different biological processes affecting different systems with the human body. For example, amylin binding can affect the immune system, the central nervous system, and the satiation system in the brain (i.e., the area postrema). There are two required post-translational modifications of amylin in order for the ligand to have any bioactivity: (1) <scene name='10/1037495/C-term_amide/2'>amidation of the C-terminus</scene> and (2) a <scene name='10/1037495/Amylin_disulfide_bond2/4'>disulfide bond</scene> between C2 and C7. | ||
| Line 23: | Line 23: | ||
There are <scene name='10/1037496/Amylin_2hbonds/4'>two conserved hydrogen bonds between the CTR and the amylin N-terminus loop</scene>. These bonds contribute to the functional phenotype of AMYR and also causes the end of the amylin ligand to be held in a flipped up position. The disulfide bonds between C2 and C7 also inhibit the end of Amylin from waving around freely, and it prevents amylin from aggregation. | There are <scene name='10/1037496/Amylin_2hbonds/4'>two conserved hydrogen bonds between the CTR and the amylin N-terminus loop</scene>. These bonds contribute to the functional phenotype of AMYR and also causes the end of the amylin ligand to be held in a flipped up position. The disulfide bonds between C2 and C7 also inhibit the end of Amylin from waving around freely, and it prevents amylin from aggregation. | ||
| - | === Binding Site Residues | + | == 12 Angstrom Calcitonin Shift == |
| + | When the RAMP binds to the CTR, the CTR undergoes a conformational change, or shift. The extracellular region of the calcitonin subunit, specifically in the C-alpha loop 5, of AMYR will undergo a 12 angstrom shift upwards. This distance shift is measured specifically from a glutamate residue, E123. This shift in the CTR facilitates the packing of the CTR extracellular domain with the antiparallel triple helix conformation of the RAMP extracellular domain. | ||
| + | |||
| + | ***** ADD PICTURE HERE OF SHIFT******** | ||
| + | |||
| + | There is little movement in the N-terminal location and midregion of the CTR when RAMP binds. This is because there is an unstructured and flexible "hinge" region between the extracellular domain and the core-binding domains, allowing these domains to remain in their original location from before RAMP binds to the CTR. | ||
| + | |||
| + | == Binding Site Residues == | ||
There are water molecules present in the binding site between amylin and the calcitonin receptor that support the ligand-receptor interaction. Some water molecules interact with the amylin ligand and create water-bridged Hydrogen bonds between different ligand residues, such as the <scene name='10/1037495/Water_1_ver3/4'>water-bridged Hydrogen bond between the main chains of T6 and T9</scene>. Other water molecules create <scene name='10/1037496/Water_receptor/1'>water-bridged Hydrogen bonds between residues of the calcitonin receptor</scene>. The water molecules are present in the empty space located in the ligand binding site, and they are hypothesized to stabilize the active conformation of the calcitonin receptor when amylin is bound. Substitutions of polar residues involved with the water-bridged Hydrogen bond network to nonpolar residues causes a decrease in potency and affinity of amylin to the calcitonin receptor. (REFERENCE NEEDED HERE******) | There are water molecules present in the binding site between amylin and the calcitonin receptor that support the ligand-receptor interaction. Some water molecules interact with the amylin ligand and create water-bridged Hydrogen bonds between different ligand residues, such as the <scene name='10/1037495/Water_1_ver3/4'>water-bridged Hydrogen bond between the main chains of T6 and T9</scene>. Other water molecules create <scene name='10/1037496/Water_receptor/1'>water-bridged Hydrogen bonds between residues of the calcitonin receptor</scene>. The water molecules are present in the empty space located in the ligand binding site, and they are hypothesized to stabilize the active conformation of the calcitonin receptor when amylin is bound. Substitutions of polar residues involved with the water-bridged Hydrogen bond network to nonpolar residues causes a decrease in potency and affinity of amylin to the calcitonin receptor. (REFERENCE NEEDED HERE******) | ||
| - | + | == G Protein Activation == | |
There are extensive nonpolar, <scene name='10/1037496/H2ophobic_interactions/2'>hydrophobic interactions between the G protein Gα subunit and the CTR 2nd intracellular loop</scene>. Val322 and Leu323 of the CTR 2nd intracellular loop makes nonpolar interactions with Ile248 and Val249 back to itself as well as Leu388 of the Gα subunit. These interactions stabilize the calcitonin receptor, which activates the G-protein to give amylin its biological role. The <scene name='10/1037495/G_protein_interaction/4'>CTR 3rd intracellular loop interacts with the Gα subunit through Hydrogen bonding</scene>. Arg180 of the 3rd intracellular loop of the calcitonin receptor makes a crucial hydrogen bond to Gln384 of the Gα subunit. This interaction has been shown to maintain the biological function of amylin through cell signaling (REFERENCE NEEDED HERE*******). These interactions between the second and third intracellular loops of the calcitonin receptor stabilize the conformational states of the G-protein to create a more rigid domain with the nanobody. | There are extensive nonpolar, <scene name='10/1037496/H2ophobic_interactions/2'>hydrophobic interactions between the G protein Gα subunit and the CTR 2nd intracellular loop</scene>. Val322 and Leu323 of the CTR 2nd intracellular loop makes nonpolar interactions with Ile248 and Val249 back to itself as well as Leu388 of the Gα subunit. These interactions stabilize the calcitonin receptor, which activates the G-protein to give amylin its biological role. The <scene name='10/1037495/G_protein_interaction/4'>CTR 3rd intracellular loop interacts with the Gα subunit through Hydrogen bonding</scene>. Arg180 of the 3rd intracellular loop of the calcitonin receptor makes a crucial hydrogen bond to Gln384 of the Gα subunit. This interaction has been shown to maintain the biological function of amylin through cell signaling (REFERENCE NEEDED HERE*******). These interactions between the second and third intracellular loops of the calcitonin receptor stabilize the conformational states of the G-protein to create a more rigid domain with the nanobody. | ||
| - | + | ||
== Biological Relevance == | == Biological Relevance == | ||
Revision as of 19:19, 24 April 2024
Amylin Receptor (AMYR)
| |||||||||||
References
- ↑ Cao J, Belousoff MJ, Liang YL, Johnson RM, Josephs TM, Fletcher MM, Christopoulos A, Hay DL, Danev R, Wootten D, Sexton PM. A structural basis for amylin receptor phenotype. Science. 2022 Mar 25;375(6587):eabm9609. PMID:35324283 doi:10.1126/science.abm9609
- ↑ Ransey E, Paredes E, Dey SK, Das SR, Heroux A, Macbeth MR. Crystal structure of the Entamoeba histolytica RNA lariat debranching enzyme EhDbr1 reveals a catalytic Zn(2+) /Mn(2+) heterobinucleation. FEBS Lett. 2017 Jul;591(13):2003-2010. doi: 10.1002/1873-3468.12677. Epub 2017, Jun 14. PMID:28504306 doi:http://dx.doi.org/10.1002/1873-3468.12677
Student Contributors
- Kylie Blake
- Natalie Link
- Jaelin Lunato