This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
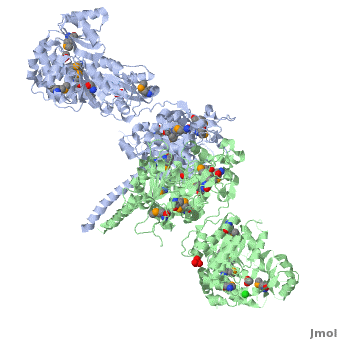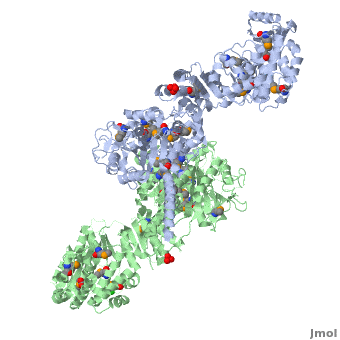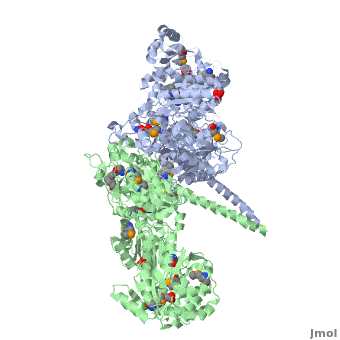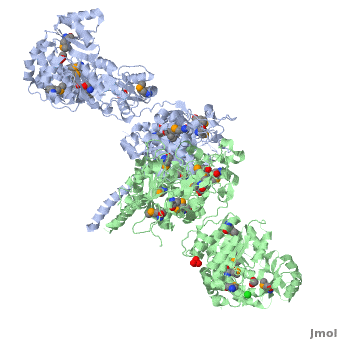
6-deoxyerythronolide B synthase (DEBS)
From Proteopedia
(Difference between revisions)
| Line 8: | Line 8: | ||
[[Image:DEBS.png|left|500px|thumb|6-deoxyerythronolide B synthase (DEBS)]] | [[Image:DEBS.png|left|500px|thumb|6-deoxyerythronolide B synthase (DEBS)]] | ||
{{Clear}} | {{Clear}} | ||
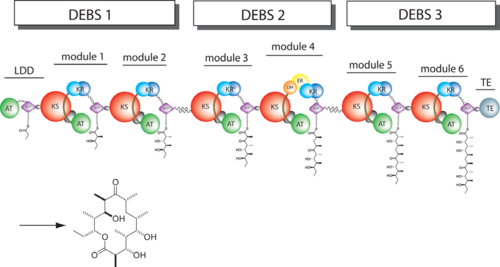
| - | The '''6-deoxyerythronolide B synthase''' (DEBS), which catalyzes the formation of 6-dEB, consists of three large subunits, DEBS1, DEBS2 and DEBS3, each containing two modules and above 300 kD in size. There are 2 domains in the N-terminal loading module, responsible for priming the synthase with a proprionate starter unit, and 26 domains in the six extender modules, Each extender module contains at least three essential domains: a ketosynthase (KS), an acyl transferase (AT) and an acyl carrier protein (ACP). In detail, the AT domain selects the appropriate carbon extender unit and transfers the units from acyl-CoA onto the phosphopantetheine arm of ACP. The KSdomain accepts the polyketide chain from the previous module and catalyzes chain elongation reaction by adding an ACP-bound extender unit through decarboxylative condensation. | + | The '''6-deoxyerythronolide B synthase''' (DEBS), which catalyzes the formation of 6-dEB, consists of three large subunits, DEBS1, DEBS2 and DEBS3, each containing two modules and above 300 kD in size. There are 2 domains in the N-terminal loading module, responsible for priming the synthase with a proprionate starter unit, and 26 domains in the six extender modules, Each extender module contains at least three essential domains: a ketosynthase (KS), an acyl transferase (AT) and an acyl carrier protein (ACP). In detail, the AT domain selects the appropriate carbon extender unit and transfers the units from acyl-CoA onto the phosphopantetheine arm of ACP. The KSdomain accepts the polyketide chain from the previous module and catalyzes chain elongation reaction by adding an ACP-bound extender unit through decarboxylative condensation.<br /> |
| + | |||
| + | '''DEBS1''' is acylated by a diketide at module 2<ref>PMID:8952473</ref>.<br /> | ||
| + | '''DEBS3''' carries out the final two cycles in the synthesis of 6-dEB<ref>PMID:9538011</ref>. | ||
After the extender unit is added, it can be further processed by optional tailoring domains, including ketoreductases (KRs), dehydratases (DHs), and enoyl reductases (ERs), to yield a hydroxyl, enoyl, or methylene group at the beta-position. Finally, the thioesterase (TE) domain that located at the C-terminus of DEBS module 6 promotes the macrocyclization event which releases the final product, 6-dEB. <ref>PMID:17328673</ref> | After the extender unit is added, it can be further processed by optional tailoring domains, including ketoreductases (KRs), dehydratases (DHs), and enoyl reductases (ERs), to yield a hydroxyl, enoyl, or methylene group at the beta-position. Finally, the thioesterase (TE) domain that located at the C-terminus of DEBS module 6 promotes the macrocyclization event which releases the final product, 6-dEB. <ref>PMID:17328673</ref> | ||
Revision as of 09:51, 19 May 2024
| |||||||||||
References
- ↑ Tsukamoto N, Chuck JA, Luo G, Kao CM, Khosla C, Cane DE. 6-deoxyerythronolide B synthase 1 is specifically acylated by a diketide intermediate at the beta-ketoacyl-acyl carrier protein synthase domain of module 2. Biochemistry. 1996 Dec 3;35(48):15244-8. PMID:8952473 doi:10.1021/bi961972f
- ↑ Jacobsen JR, Cane DE, Khosla C. Spontaneous priming of a downstream module in 6-deoxyerythronolide B synthase leads to polyketide biosynthesis. Biochemistry. 1998 Apr 7;37(14):4928-34. PMID:9538011 doi:10.1021/bi9729920
- ↑ Khosla C, Tang Y, Chen AY, Schnarr NA, Cane DE. Structure and mechanism of the 6-deoxyerythronolide B synthase. Annu Rev Biochem. 2007;76:195-221. PMID:17328673 doi:http://dx.doi.org/10.1146/annurev.biochem.76.053105.093515
Additional Resources
For additional information, see: Cancer
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Tsung-Yi Lin, Alexander Berchansky, Lawrence Sheringham Borketey, Joel L. Sussman, Jon Amoroso, David Canner, Jaime Prilusky