Sandbox Reserved 1846
From Proteopedia
(Difference between revisions)
| Line 31: | Line 31: | ||
== Mutation Sites of Interest == | == Mutation Sites of Interest == | ||
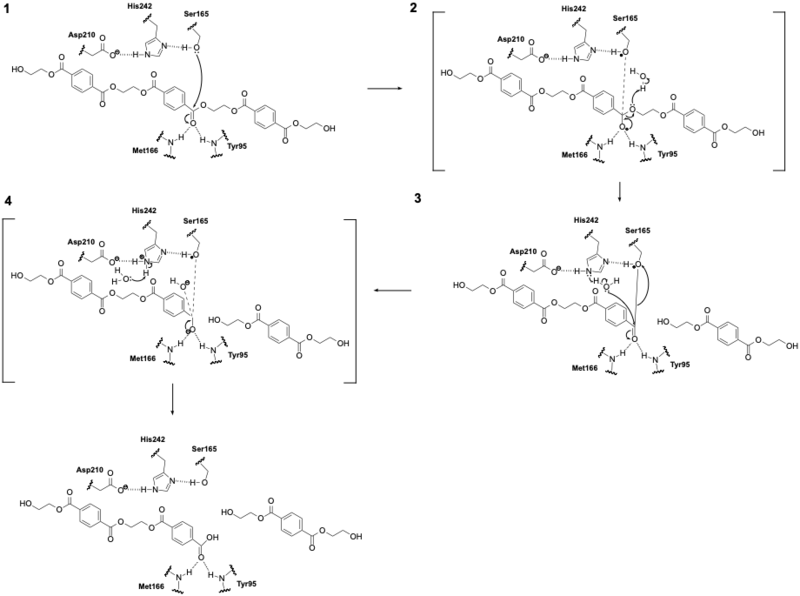
| + | To improve the catalytic activity and thermostability of LCC, Tournier et al. (2020) used structure-guided enzyme engineering based on the crystal structure of LCC bound to a model PET substrate. Using molecular docking and enzyme–substrate contact analysis, the researchers identified 15 residues in the first contact shell surrounding the substrate-binding groove. Of these, 11 positions were selected for saturation mutagenesis to determine how mutations could affect PET depolymerization. These sites were chosen for their interactions with the PET-like ligand or their proximity to the active site. Highly conserved residues essential for catalysis or structural stability were excluded. | ||
| + | From this screen, two mutations at Phe243 (F243I and F243W) were shown to improve catalytic activity by optimizing substrate positioning within the groove. To increase thermostability, the authors targeted a region of the enzyme that is structurally analogous to known divalent metal-binding sites in other cutinases. Instead of using stabilizing ions, which could complicate industrial processes, they engineered a disulfide bridge by mutating Asp238 and Ser283 to Cys residues (D238C/S283C). Additional mutations were selected based on thermostability screening. Among these, Y127G improved the melting point without reducing activity. | ||
| + | These mutations were combined to create multi-mutant LCC variants with improved activity and thermostability. The two most successful variants were: | ||
| + | ICCG: F243I / D238C / S283C / Y127G | ||
| + | WCCG: F243W / D238C / S283C / Y127G | ||
| + | It was frequently included in the best-performing multi-mutant variants. Other stabilizing mutations, such as T96M, N246D, and N246M, were also tested but are not included in this page's protein model. These were excluded either because they were not part of the top-performing mutant (ICCG), or because their effects were less impactful individually and therefore omitted for clarity. | ||
| + | |||
| + | |||
=== F243 === | === F243 === | ||
Revision as of 01:46, 15 April 2025
| This Sandbox is Reserved from March 18 through September 1, 2025 for use in the course CH462 Biochemistry II taught by R. Jeremy Johnson and Mark Macbeth at the Butler University, Indianapolis, USA. This reservation includes Sandbox Reserved 1828 through Sandbox Reserved 1846. |
To get started:
More help: Help:Editing |
Leaf Branch Compost Cutinase
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 Tournier V, Topham CM, Gilles A, David B, Folgoas C, Moya-Leclair E, Kamionka E, Desrousseaux ML, Texier H, Gavalda S, Cot M, Guemard E, Dalibey M, Nomme J, Cioci G, Barbe S, Chateau M, Andre I, Duquesne S, Marty A. An engineered PET depolymerase to break down and recycle plastic bottles. Nature. 2020 Apr;580(7802):216-219. doi: 10.1038/s41586-020-2149-4. Epub 2020 Apr, 8. PMID:32269349 doi:http://dx.doi.org/10.1038/s41586-020-2149-4
- ↑ 2.0 2.1 2.2 2.3 2.4 Sui B, Wang T, Fang J, Hou Z, Shu T, Lu Z, Liu F, Zhu Y. Recent advances in the biodegradation of polyethylene terephthalate with cutinase-like enzymes. Front Microbiol. 2023 Oct 2;14:1265139. PMID:37849919 doi:10.3389/fmicb.2023.1265139
- ↑ Ueda H, Tabata J, Seshime Y, Masaki K, Sameshima-Yamashita Y, Kitamoto H. Cutinase-like biodegradable plastic-degrading enzymes from phylloplane yeasts have cutinase activity. Biosci Biotechnol Biochem. 2021 Jul 23;85(8):1890-1898. PMID:34160605 doi:10.1093/bbb/zbab113
- ↑ Kolattukudy PE. Biopolyester membranes of plants: cutin and suberin. Science. 1980 May 30;208(4447):990-1000. PMID:17779010 doi:10.1126/science.208.4447.990
- ↑ 5.0 5.1 5.2 5.3 5.4 Khairul Anuar NFS, Huyop F, Ur-Rehman G, Abdullah F, Normi YM, Sabullah MK, Abdul Wahab R. An Overview into Polyethylene Terephthalate (PET) Hydrolases and Efforts in Tailoring Enzymes for Improved Plastic Degradation. Int J Mol Sci. 2022 Oct 20;23(20):12644. PMID:36293501 doi:10.3390/ijms232012644
- ↑ 6.0 6.1 Burgin T, Pollard BC, Knott BC, Mayes HB, Crowley MF, McGeehan JE, Beckham GT, Woodcock HL. The reaction mechanism of the Ideonella sakaiensis PETase enzyme. Commun Chem. 2024 Mar 27;7(1):65. PMID:38538850 doi:10.1038/s42004-024-01154-x
- ↑ 7.0 7.1 Stevensen J, Janatunaim RZ, Ratnaputri AH, Aldafa SH, Rudjito RR, Saputro DH, Suhandono S, Putri RM, Aditama R, Fibriani A. Thermostability and Activity Improvements of PETase from Ideonella sakaiensis. ACS Omega. 2025 Feb 12;10(7):6385-6395. PMID:40028137 doi:10.1021/acsomega.4c05142
- ↑ Yoshida S, Hiraga K, Takehana T, Taniguchi I, Yamaji H, Maeda Y, Toyohara K, Miyamoto K, Kimura Y, Oda K. A bacterium that degrades and assimilates poly(ethylene terephthalate). Science. 2016 Mar 11;351(6278):1196-9. doi: 10.1126/science.aad6359. PMID:26965627 doi:http://dx.doi.org/10.1126/science.aad6359
- ↑ Landrigan PJ, Stegeman JJ, Fleming LE, Allemand D, Anderson DM, Backer LC, Brucker-Davis F, Chevalier N, Corra L, Czerucka D, Bottein MD, Demeneix B, Depledge M, Deheyn DD, Dorman CJ, Fénichel P, Fisher S, Gaill F, Galgani F, Gaze WH, Giuliano L, Grandjean P, Hahn ME, Hamdoun A, Hess P, Judson B, Laborde A, McGlade J, Mu J, Mustapha A, Neira M, Noble RT, Pedrotti ML, Reddy C, Rocklöv J, Scharler UM, Shanmugam H, Taghian G, van de Water JAJM, Vezzulli L, Weihe P, Zeka A, Raps H, Rampal P. Human Health and Ocean Pollution. Ann Glob Health. 2020 Dec 3;86(1):151. PMID:33354517 doi:10.5334/aogh.2831
- ↑ Jambeck JR, Geyer R, Wilcox C, Siegler TR, Perryman M, Andrady A, Narayan R, Law KL. Marine pollution. Plastic waste inputs from land into the ocean. Science. 2015 Feb 13;347(6223):768-71. PMID:25678662 doi:10.1126/science.1260352
Student Contributors
Ashley Callaghan Rebecca Hoff Simone McCowan