This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
6pax
From Proteopedia
(New page: 200px<br /> <applet load="6pax" size="450" color="white" frame="true" align="right" spinBox="true" caption="6pax, resolution 2.50Å" /> '''CRYSTAL STRUCTURE O...) |
|||
| Line 1: | Line 1: | ||
| - | [[Image:6pax.gif|left|200px]]<br /> | + | [[Image:6pax.gif|left|200px]]<br /><applet load="6pax" size="350" color="white" frame="true" align="right" spinBox="true" |
| - | <applet load="6pax" size=" | + | |
caption="6pax, resolution 2.50Å" /> | caption="6pax, resolution 2.50Å" /> | ||
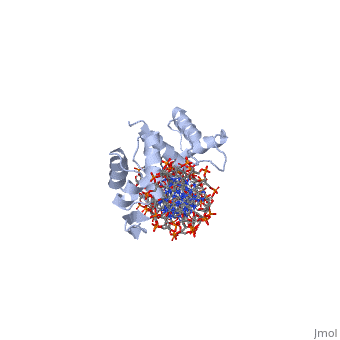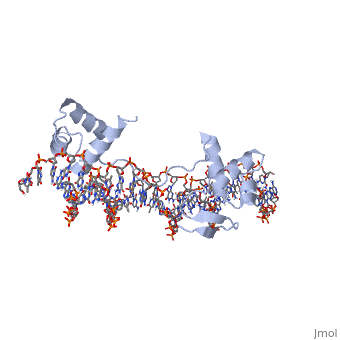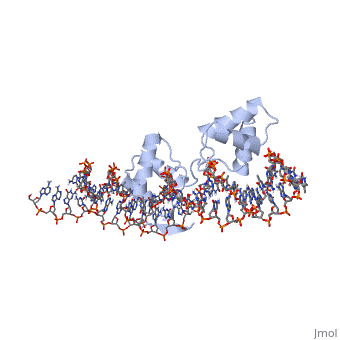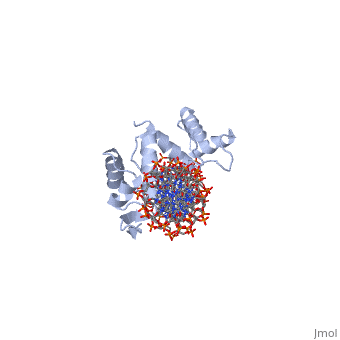
'''CRYSTAL STRUCTURE OF THE HUMAN PAX-6 PAIRED DOMAIN-DNA COMPLEX REVEALS A GENERAL MODEL FOR PAX PROTEIN-DNA INTERACTIONS'''<br /> | '''CRYSTAL STRUCTURE OF THE HUMAN PAX-6 PAIRED DOMAIN-DNA COMPLEX REVEALS A GENERAL MODEL FOR PAX PROTEIN-DNA INTERACTIONS'''<br /> | ||
==Overview== | ==Overview== | ||
| - | Pax6, a transcription factor containing the bipartite paired DNA-binding | + | Pax6, a transcription factor containing the bipartite paired DNA-binding domain, has critical roles in development of the eye, nose, pancreas, and central nervous system. The 2.5 A structure of the human Pax6 paired domain with its optimal 26-bp site reveals extensive DNA contacts from the amino-terminal subdomain, the linker region, and the carboxy-terminal subdomain. The Pax6 structure not only confirms the docking arrangement of the amino-terminal subdomain as seen in cocrystals of the Drosophila Prd Pax protein, but also reveals some interesting differences in this region and helps explain the sequence specificity of paired domain-DNA recognition. In addition, this structure gives the first detailed information about how the paired linker region and carboxy-terminal subdomain contact DNA. The extended linker makes minor groove contacts over an 8-bp region, and the carboxy-terminal helix-turn-helix unit makes base contacts in the major groove. The structure and docking arrangement of the carboxy-terminal subdomain of Pax6 is remarkably similar to that of the amino-terminal subdomain, and there is an approximate twofold symmetry axis relating the polypeptide backbones of these two helix-turn-helix units. Our structure of the Pax6 paired domain-DNA complex provides a framework for understanding paired domain-DNA interactions, for analyzing mutations that map in the linker and carboxy-terminal regions of the paired domain, and for modeling protein-protein interactions of the Pax family proteins. |
==Disease== | ==Disease== | ||
| Line 11: | Line 10: | ||
==About this Structure== | ==About this Structure== | ||
| - | 6PAX is a [http://en.wikipedia.org/wiki/Single_protein Single protein] structure of sequence from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens]. Full crystallographic information is available from [http:// | + | 6PAX is a [http://en.wikipedia.org/wiki/Single_protein Single protein] structure of sequence from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=6PAX OCA]. |
==Reference== | ==Reference== | ||
| Line 17: | Line 16: | ||
[[Category: Homo sapiens]] | [[Category: Homo sapiens]] | ||
[[Category: Single protein]] | [[Category: Single protein]] | ||
| - | [[Category: Epstein, J | + | [[Category: Epstein, J A.]] |
| - | [[Category: Maas, R | + | [[Category: Maas, R L.]] |
| - | [[Category: Pabo, C | + | [[Category: Pabo, C O.]] |
| - | [[Category: Rould, M | + | [[Category: Rould, M A.]] |
| - | [[Category: Xu, H | + | [[Category: Xu, H E.]] |
[[Category: Xu, W.]] | [[Category: Xu, W.]] | ||
[[Category: paired domain]] | [[Category: paired domain]] | ||
| Line 28: | Line 27: | ||
[[Category: transcription]] | [[Category: transcription]] | ||
| - | ''Page seeded by [http:// | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Feb 21 19:16:40 2008'' |
Revision as of 17:16, 21 February 2008
|
CRYSTAL STRUCTURE OF THE HUMAN PAX-6 PAIRED DOMAIN-DNA COMPLEX REVEALS A GENERAL MODEL FOR PAX PROTEIN-DNA INTERACTIONS
Contents |
Overview
Pax6, a transcription factor containing the bipartite paired DNA-binding domain, has critical roles in development of the eye, nose, pancreas, and central nervous system. The 2.5 A structure of the human Pax6 paired domain with its optimal 26-bp site reveals extensive DNA contacts from the amino-terminal subdomain, the linker region, and the carboxy-terminal subdomain. The Pax6 structure not only confirms the docking arrangement of the amino-terminal subdomain as seen in cocrystals of the Drosophila Prd Pax protein, but also reveals some interesting differences in this region and helps explain the sequence specificity of paired domain-DNA recognition. In addition, this structure gives the first detailed information about how the paired linker region and carboxy-terminal subdomain contact DNA. The extended linker makes minor groove contacts over an 8-bp region, and the carboxy-terminal helix-turn-helix unit makes base contacts in the major groove. The structure and docking arrangement of the carboxy-terminal subdomain of Pax6 is remarkably similar to that of the amino-terminal subdomain, and there is an approximate twofold symmetry axis relating the polypeptide backbones of these two helix-turn-helix units. Our structure of the Pax6 paired domain-DNA complex provides a framework for understanding paired domain-DNA interactions, for analyzing mutations that map in the linker and carboxy-terminal regions of the paired domain, and for modeling protein-protein interactions of the Pax family proteins.
Disease
Known diseases associated with this structure: Aniridia, type II OMIM:[607108], Cataract, congenital, with late-onset corneal dystrophy OMIM:[607108], Coloboma, ocular OMIM:[607108], Ectopia pupillae OMIM:[607108], Eye anomalies, multiplex OMIM:[607108], Foveal hypoplasia, isolated OMIM:[607108], Keratitis OMIM:[607108], Morning glory disc anomaly OMIM:[607108], Optic nerve hypoplasia/aplasia OMIM:[607108], Peters anomaly OMIM:[607108]
About this Structure
6PAX is a Single protein structure of sequence from Homo sapiens. Full crystallographic information is available from OCA.
Reference
Crystal structure of the human Pax6 paired domain-DNA complex reveals specific roles for the linker region and carboxy-terminal subdomain in DNA binding., Xu HE, Rould MA, Xu W, Epstein JA, Maas RL, Pabo CO, Genes Dev. 1999 May 15;13(10):1263-75. PMID:10346815
Page seeded by OCA on Thu Feb 21 19:16:40 2008