This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1nfk
From Proteopedia
(New page: 200px<br /><applet load="1nfk" size="450" color="white" frame="true" align="right" spinBox="true" caption="1nfk, resolution 2.300Å" /> '''STRUCTURE OF THE NU...) |
|||
| Line 1: | Line 1: | ||
| - | [[Image:1nfk.jpg|left|200px]]<br /><applet load="1nfk" size=" | + | [[Image:1nfk.jpg|left|200px]]<br /><applet load="1nfk" size="350" color="white" frame="true" align="right" spinBox="true" |
caption="1nfk, resolution 2.300Å" /> | caption="1nfk, resolution 2.300Å" /> | ||
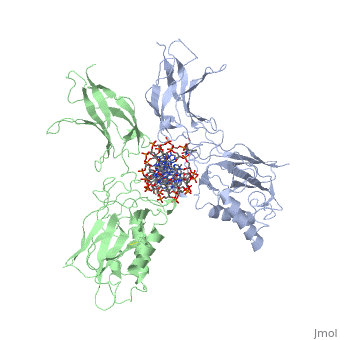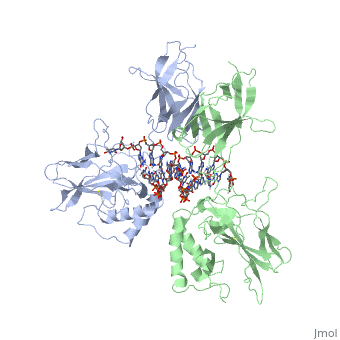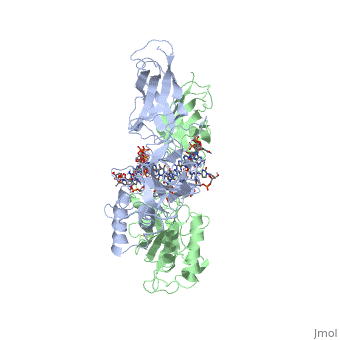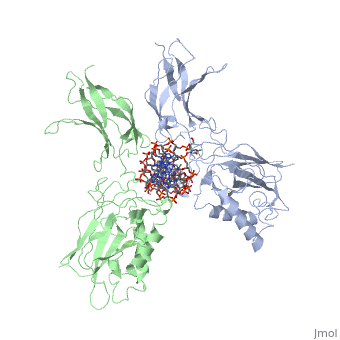
'''STRUCTURE OF THE NUCLEAR FACTOR KAPPA-B (NF-KB) P50 HOMODIMER'''<br /> | '''STRUCTURE OF THE NUCLEAR FACTOR KAPPA-B (NF-KB) P50 HOMODIMER'''<br /> | ||
==Overview== | ==Overview== | ||
| - | The 2.3-A crystal structure of the transcription factor NK-kappa B p50 | + | The 2.3-A crystal structure of the transcription factor NK-kappa B p50 homodimer bound to a palindromic kappa B site reveals that the Rel homology region folds into two distinct domains, similar to those in the immunoglobulin superfamily. The p50 dimer envelopes an undistorted B-DNA helix, making specific contacts along the 10-base-pair kappa B recognition site mainly through loops connecting secondary structure elements in both domains. The carboxy-terminal domains form a dimerization interface between beta-sheets using residues that are strongly conserved in the Rel family. |
==About this Structure== | ==About this Structure== | ||
| - | 1NFK is a [http://en.wikipedia.org/wiki/Single_protein Single protein] structure of sequence from [http://en.wikipedia.org/wiki/Mus_musculus Mus musculus]. Full crystallographic information is available from [http:// | + | 1NFK is a [http://en.wikipedia.org/wiki/Single_protein Single protein] structure of sequence from [http://en.wikipedia.org/wiki/Mus_musculus Mus musculus]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1NFK OCA]. |
==Reference== | ==Reference== | ||
| Line 13: | Line 13: | ||
[[Category: Mus musculus]] | [[Category: Mus musculus]] | ||
[[Category: Single protein]] | [[Category: Single protein]] | ||
| - | [[Category: Duyne, G | + | [[Category: Duyne, G Van.]] |
[[Category: Ghosh, G.]] | [[Category: Ghosh, G.]] | ||
[[Category: Ghosh, S.]] | [[Category: Ghosh, S.]] | ||
| - | [[Category: Sigler, P | + | [[Category: Sigler, P B.]] |
[[Category: complex (transcription factor/dna)]] | [[Category: complex (transcription factor/dna)]] | ||
[[Category: nf-kb p50]] | [[Category: nf-kb p50]] | ||
| - | ''Page seeded by [http:// | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Feb 21 14:05:34 2008'' |
Revision as of 12:05, 21 February 2008
|
STRUCTURE OF THE NUCLEAR FACTOR KAPPA-B (NF-KB) P50 HOMODIMER
Overview
The 2.3-A crystal structure of the transcription factor NK-kappa B p50 homodimer bound to a palindromic kappa B site reveals that the Rel homology region folds into two distinct domains, similar to those in the immunoglobulin superfamily. The p50 dimer envelopes an undistorted B-DNA helix, making specific contacts along the 10-base-pair kappa B recognition site mainly through loops connecting secondary structure elements in both domains. The carboxy-terminal domains form a dimerization interface between beta-sheets using residues that are strongly conserved in the Rel family.
About this Structure
1NFK is a Single protein structure of sequence from Mus musculus. Full crystallographic information is available from OCA.
Reference
Structure of NF-kappa B p50 homodimer bound to a kappa B site., Ghosh G, van Duyne G, Ghosh S, Sigler PB, Nature. 1995 Jan 26;373(6512):303-10. PMID:7530332
Page seeded by OCA on Thu Feb 21 14:05:34 2008