This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox2qc8
From Proteopedia
(Difference between revisions)
| Line 11: | Line 11: | ||
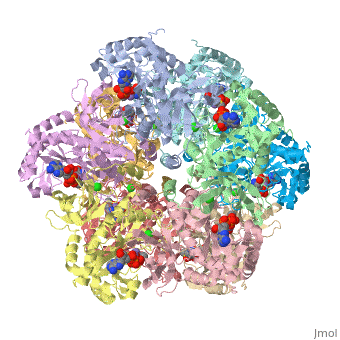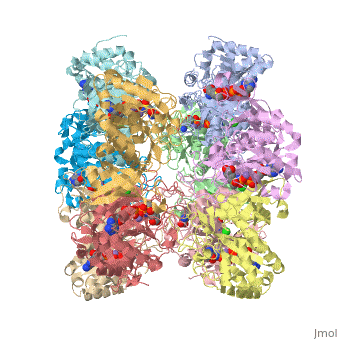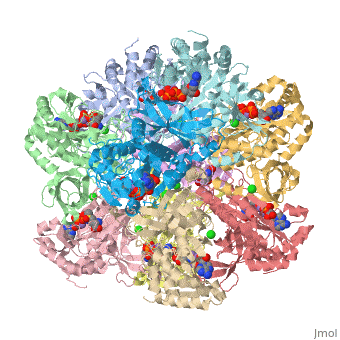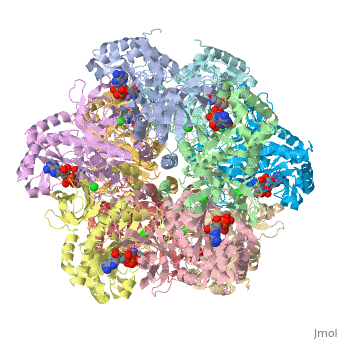
Glutamine synthetase is composed of 10 <scene name='Sandbox2qc8/Secondary_structure/1'>identical subunits</scene>. | Glutamine synthetase is composed of 10 <scene name='Sandbox2qc8/Secondary_structure/1'>identical subunits</scene>. | ||
| + | |||
| + | Each subunit has an exposed NH2 terminus and buried COOH terminus. <ref>Yamashita, M., et al.,Refined Atomic Model of Glutamine Synthetase at 3.5A Resolution, The Journal of Biological Chemistry, 1989, 17681-17690.</ref> | ||
Each subunit is composed predominantly of 12 <scene name='Sandbox2qc8/Subunit_alpha/1'>alpha helices</scene> and 12 | Each subunit is composed predominantly of 12 <scene name='Sandbox2qc8/Subunit_alpha/1'>alpha helices</scene> and 12 | ||
| Line 17: | Line 19: | ||
The beta sheets are arranged into two separate partial beta barrels, one of which encompasses the ligand complex. | The beta sheets are arranged into two separate partial beta barrels, one of which encompasses the ligand complex. | ||
| - | The active site within the secondary structure can be called a "bifunnel," providing access to ATP and glutamate at opposing ends.<ref>Eisenberg, D., et al, Structure-function relationships of glutamine synthetases, Biochimica et Biophysica Acta 1477 (2000), 122-145.</ref | + | The active site within the secondary structure can be called a "bifunnel," providing access to ATP and glutamate at opposing ends.<ref>Eisenberg, D., et al., Structure-function relationships of glutamine synthetases, Biochimica et Biophysica Acta 1477 (2000), 122-145.</ref> |
Revision as of 02:59, 8 December 2008
| |||||||||
| 2qc8, resolution 2.60Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligands: | , , , | ||||||||
| Gene: | GLUL, GLNS (Homo sapiens) | ||||||||
| Activity: | Glutamate--ammonia ligase, with EC number 6.3.1.2 | ||||||||
| Related: | 2ojw | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, RCSB, PDBsum | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Click here to view .
Click here to view .
Click here to view .
Click here to view .
Glutamine synthetase is composed of 10 .
Each subunit has an exposed NH2 terminus and buried COOH terminus. [1]
Each subunit is composed predominantly of 12 and 12 , as well as a .
The beta sheets are arranged into two separate partial beta barrels, one of which encompasses the ligand complex.
The active site within the secondary structure can be called a "bifunnel," providing access to ATP and glutamate at opposing ends.[2]
The ligands present are Cl, Mn, Adenosine Diphosphate, and L-Methionine-S-Sulfoximine Phosphate.