This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
2bo5
From Proteopedia
(New page: 200px<br /><applet load="2bo5" size="450" color="white" frame="true" align="right" spinBox="true" caption="2bo5" /> '''BOVINE OLIGOMYCIN SENSITIVITY CONFERRAL PROT...) |
|||
| Line 1: | Line 1: | ||
| - | [[Image:2bo5.gif|left|200px]]<br /><applet load="2bo5" size=" | + | [[Image:2bo5.gif|left|200px]]<br /><applet load="2bo5" size="350" color="white" frame="true" align="right" spinBox="true" |
caption="2bo5" /> | caption="2bo5" /> | ||
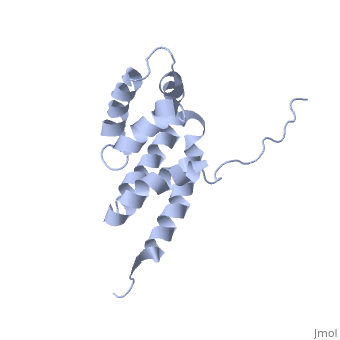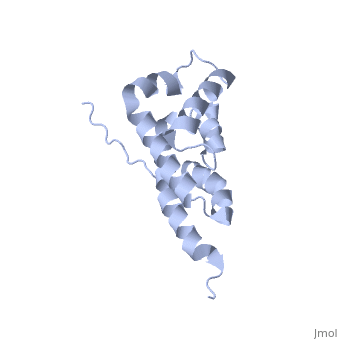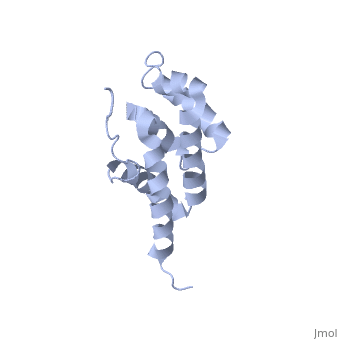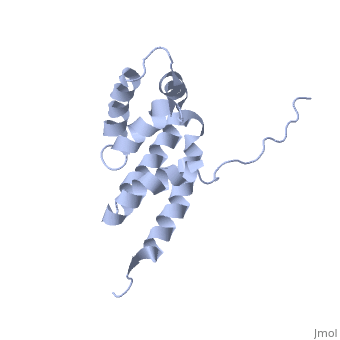
'''BOVINE OLIGOMYCIN SENSITIVITY CONFERRAL PROTEIN N-TERMINAL DOMAIN'''<br /> | '''BOVINE OLIGOMYCIN SENSITIVITY CONFERRAL PROTEIN N-TERMINAL DOMAIN'''<br /> | ||
==Overview== | ==Overview== | ||
| - | The peripheral stalk of ATP synthase holds the alpha3beta3 catalytic | + | The peripheral stalk of ATP synthase holds the alpha3beta3 catalytic subcomplex stationary against the torque of the rotating central stalk. In bovine mitochondria, the N-terminal domain of the oligomycin sensitivity conferral protein (OSCP-NT; residues 1-120) anchors one end of the peripheral stalk to the N-terminal tails of one or more alpha-subunits of the F1 subcomplex. Here we present the solution structure of OSCP-NT and an NMR titration study of its interaction with peptides representing N-terminal tails of F1 alpha-subunits. The structure comprises a bundle of six alpha-helices, and its interaction site contains adjoining hydrophobic surfaces of helices 1 and 5; residues in the region 1-8 of the alpha-subunit are essential for the interaction. The OSCP-NT is similar to the N-terminal domain of the delta-subunit from Escherichia coli ATP synthase (delta-NT), except that their surface charges differ (basic and acidic, respectively). As the charges of the adjacent crown regions in their alpha3beta3 complexes are similar, the OSCP-NT and delta-NT probably do not contact the crowns extensively. The N-terminal tails of alpha-subunit tails are probably alpha-helical, and so this interface, which is essential for the rotary mechanism of the enzyme, appears to consist of helix-helix interactions. |
==About this Structure== | ==About this Structure== | ||
| - | 2BO5 is a [http://en.wikipedia.org/wiki/Single_protein Single protein] structure of sequence from [http://en.wikipedia.org/wiki/Bos_taurus Bos taurus]. Active as [http://en.wikipedia.org/wiki/H(+)-transporting_two-sector_ATPase H(+)-transporting two-sector ATPase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=3.6.3.14 3.6.3.14] Full crystallographic information is available from [http:// | + | 2BO5 is a [http://en.wikipedia.org/wiki/Single_protein Single protein] structure of sequence from [http://en.wikipedia.org/wiki/Bos_taurus Bos taurus]. Active as [http://en.wikipedia.org/wiki/H(+)-transporting_two-sector_ATPase H(+)-transporting two-sector ATPase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=3.6.3.14 3.6.3.14] Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2BO5 OCA]. |
==Reference== | ==Reference== | ||
| Line 14: | Line 14: | ||
[[Category: H(+)-transporting two-sector ATPase]] | [[Category: H(+)-transporting two-sector ATPase]] | ||
[[Category: Single protein]] | [[Category: Single protein]] | ||
| - | [[Category: Carbajo, R | + | [[Category: Carbajo, R J.]] |
| - | [[Category: Kellas, F | + | [[Category: Kellas, F A.]] |
| - | [[Category: Montgomery, M | + | [[Category: Montgomery, M G.]] |
[[Category: Neuhaus, D.]] | [[Category: Neuhaus, D.]] | ||
| - | [[Category: Runswick, M | + | [[Category: Runswick, M J.]] |
| - | [[Category: Walker, J | + | [[Category: Walker, J E.]] |
[[Category: alpha-subunit]] | [[Category: alpha-subunit]] | ||
[[Category: atp synthase]] | [[Category: atp synthase]] | ||
| Line 39: | Line 39: | ||
[[Category: transport]] | [[Category: transport]] | ||
| - | ''Page seeded by [http:// | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Feb 21 16:39:51 2008'' |
Revision as of 14:39, 21 February 2008
|
BOVINE OLIGOMYCIN SENSITIVITY CONFERRAL PROTEIN N-TERMINAL DOMAIN
Overview
The peripheral stalk of ATP synthase holds the alpha3beta3 catalytic subcomplex stationary against the torque of the rotating central stalk. In bovine mitochondria, the N-terminal domain of the oligomycin sensitivity conferral protein (OSCP-NT; residues 1-120) anchors one end of the peripheral stalk to the N-terminal tails of one or more alpha-subunits of the F1 subcomplex. Here we present the solution structure of OSCP-NT and an NMR titration study of its interaction with peptides representing N-terminal tails of F1 alpha-subunits. The structure comprises a bundle of six alpha-helices, and its interaction site contains adjoining hydrophobic surfaces of helices 1 and 5; residues in the region 1-8 of the alpha-subunit are essential for the interaction. The OSCP-NT is similar to the N-terminal domain of the delta-subunit from Escherichia coli ATP synthase (delta-NT), except that their surface charges differ (basic and acidic, respectively). As the charges of the adjacent crown regions in their alpha3beta3 complexes are similar, the OSCP-NT and delta-NT probably do not contact the crowns extensively. The N-terminal tails of alpha-subunit tails are probably alpha-helical, and so this interface, which is essential for the rotary mechanism of the enzyme, appears to consist of helix-helix interactions.
About this Structure
2BO5 is a Single protein structure of sequence from Bos taurus. Active as H(+)-transporting two-sector ATPase, with EC number 3.6.3.14 Full crystallographic information is available from OCA.
Reference
Structure of the F1-binding domain of the stator of bovine F1Fo-ATPase and how it binds an alpha-subunit., Carbajo RJ, Kellas FA, Runswick MJ, Montgomery MG, Walker JE, Neuhaus D, J Mol Biol. 2005 Aug 26;351(4):824-38. PMID:16045926
Page seeded by OCA on Thu Feb 21 16:39:51 2008
Categories: Bos taurus | H(+)-transporting two-sector ATPase | Single protein | Carbajo, R J. | Kellas, F A. | Montgomery, M G. | Neuhaus, D. | Runswick, M J. | Walker, J E. | Alpha-subunit | Atp synthase | Beta-subunit | Binding interface | Cf(1) | Chemical shift mapping | Chemical shift perturbations | Hydrogen ion transport | Hydrolase | Ion transport | Mitochondrion | Nmr | Oscp | Peripheral stalk | Protein-protein interactions | Titration | Transit peptide | Transport