Ketosteroid Isomerase
From Proteopedia
| Line 1: | Line 1: | ||
==Ketosteroid Isomerase== | ==Ketosteroid Isomerase== | ||
| - | ==Introduction== | + | ===Introduction=== |
<applet load='1ISK' size='400' frame='true' align='right' caption='Ketosteroid Isomerase (KSI; PDB 1ISK)' scene='User:Laura_M._Haynes/Sandbox_1/Ksi/4' /> | <applet load='1ISK' size='400' frame='true' align='right' caption='Ketosteroid Isomerase (KSI; PDB 1ISK)' scene='User:Laura_M._Haynes/Sandbox_1/Ksi/4' /> | ||
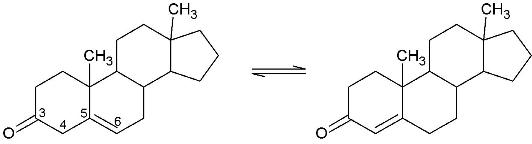
<scene name='User:Laura_M._Haynes/Sandbox_1/Ksi/4'>Ketosteroid isomerase</scene> (KSI, EC#5.3.3.1) is an enzyme that catalyzes the isomerization of 3-oxo-Δ<sup>5</sup> ketosteroids to their hormonally active Δ<sup>4</sup>-conjugated isomers, as illustrated below.<ref name="Pollack">PMID:15381400</ref>, <ref name="Talalay">PMID:7358699</ref> | <scene name='User:Laura_M._Haynes/Sandbox_1/Ksi/4'>Ketosteroid isomerase</scene> (KSI, EC#5.3.3.1) is an enzyme that catalyzes the isomerization of 3-oxo-Δ<sup>5</sup> ketosteroids to their hormonally active Δ<sup>4</sup>-conjugated isomers, as illustrated below.<ref name="Pollack">PMID:15381400</ref>, <ref name="Talalay">PMID:7358699</ref> | ||
| Line 10: | Line 10: | ||
An NMR solution phase structure of KSI was solved in 1997 by Wu ''et al.''<ref name="Wu">PMID:9103200 </ref> allowing greater insight into the mechanism of this intriguing enzyme. | An NMR solution phase structure of KSI was solved in 1997 by Wu ''et al.''<ref name="Wu">PMID:9103200 </ref> allowing greater insight into the mechanism of this intriguing enzyme. | ||
| - | ==Structure== | + | ===Structure=== |
Ketosteroid isomerase exits as a 28 kDa homodimeric protein, in which the two dimers related to each other via hydrophobic and electrostatic interactions.<ref name="Wu" /> Each dimer consists of a curved <scene name='User:Laura_M._Haynes/Sandbox_1/Beta_sheets/1'>beta-sheets</scene> and three <scene name='User:Laura_M._Haynes/Sandbox_1/Alpha_helices/1'>alpha-helices</scene>. These secondary structures define a conical closed barrel geometry, with one open and one closed end, and create a deep pocket in which the active site resides.<ref name="Ha" />,<ref name="Cho">PMID:9622484 </ref> This unique geometry is shared by several other proteins (scytalone dehydratase, nuclear transport factor 2, and naphthalene 1,2-dioxygenase), however, these molecules do not share functional or sequence homology. It is speculated that this unique protein structure may enable better binding of hydrophobic substrates such as steroids.<ref name="Ha" /> | Ketosteroid isomerase exits as a 28 kDa homodimeric protein, in which the two dimers related to each other via hydrophobic and electrostatic interactions.<ref name="Wu" /> Each dimer consists of a curved <scene name='User:Laura_M._Haynes/Sandbox_1/Beta_sheets/1'>beta-sheets</scene> and three <scene name='User:Laura_M._Haynes/Sandbox_1/Alpha_helices/1'>alpha-helices</scene>. These secondary structures define a conical closed barrel geometry, with one open and one closed end, and create a deep pocket in which the active site resides.<ref name="Ha" />,<ref name="Cho">PMID:9622484 </ref> This unique geometry is shared by several other proteins (scytalone dehydratase, nuclear transport factor 2, and naphthalene 1,2-dioxygenase), however, these molecules do not share functional or sequence homology. It is speculated that this unique protein structure may enable better binding of hydrophobic substrates such as steroids.<ref name="Ha" /> | ||
| Line 16: | Line 16: | ||
| - | ==Enzyme Mechanism== | + | ===Enzyme Mechanism=== |
| - | The general mechanism of the proposed reaction of ketosteroid isomerase involves the breaking of a C-H bond adjacent to a carbonyl. This is typically regarded as a difficult reaction due to instability of the intermediate; however it is observed in a number of enzyme-mediated biological reactions.<ref name="Pollack" /> In line with other biological reactions, the mechanism of KSI involves the abstraction the β-hyrdogen from the 4-position carbon resulting in the formation of an enol intermediate, which is followed by reketonization.<ref name="Pollack" />,<ref name="Ha" /> Structural and kinetic studies suggest that Asp<sup>38</sup> (numbering is that of the TI varient of KSI) serves as a general base in this reaction | + | The general mechanism of the proposed reaction of ketosteroid isomerase involves the breaking of a C-H bond adjacent to a carbonyl. This is typically regarded as a difficult reaction due to instability of the intermediate; however it is observed in a number of enzyme-mediated biological reactions.<ref name="Pollack" /> In line with other biological reactions, the mechanism of KSI involves the abstraction the β-hyrdogen from the 4-position carbon resulting in the formation of an enol intermediate, which is followed by reketonization.<ref name="Pollack" />,<ref name="Ha" /> Structural and kinetic studies suggest that Asp<sup>38</sup> (numbering is that of the TI varient of KSI) serves as a general base in this reaction as shown below. |
| - | ==Related Proteins== | ||
| - | ==Available Structures== | ||
| - | ==References== | + | Tyr<sup>14</sup> and Asp<sup>99</sup> are believed hydrogen bond to the O-3 carbonyl of the substrate steroid and stabilize reaction intermediates. Tyr<sup>14</sup> is also believed to participate in a low barrier hydrogen bond with the 3-postition oxygen of the steroid, thereby facilitating the abstraction of the β-hydrogen at the 4-position. |
| + | |||
| + | ===Related Proteins=== | ||
| + | |||
| + | ===Available Structures=== | ||
| + | |||
| + | ===References=== | ||
<references /> | <references /> | ||
Revision as of 18:31, 3 April 2010
Contents |
Ketosteroid Isomerase
Introduction
|
(KSI, EC#5.3.3.1) is an enzyme that catalyzes the isomerization of 3-oxo-Δ5 ketosteroids to their hormonally active Δ4-conjugated isomers, as illustrated below.[1], [2]
This reaction is essential in the biosynthesis of steroids in mammals where KSI is a membrane-bound complex.[3] In bacteria, however, KSI exists as a soluble protein is involves in catabolism of steroids.[3] It was first isolated in and has been extensively studied in Commamonas tetosteroni (TI), a bacteria that is capable of It is one of the most efficient known enzymes with an essentially diffusion limited rate of catalysis.[2]
An NMR solution phase structure of KSI was solved in 1997 by Wu et al.[4] allowing greater insight into the mechanism of this intriguing enzyme.
Structure
Ketosteroid isomerase exits as a 28 kDa homodimeric protein, in which the two dimers related to each other via hydrophobic and electrostatic interactions.[4] Each dimer consists of a curved and three . These secondary structures define a conical closed barrel geometry, with one open and one closed end, and create a deep pocket in which the active site resides.[3],[5] This unique geometry is shared by several other proteins (scytalone dehydratase, nuclear transport factor 2, and naphthalene 1,2-dioxygenase), however, these molecules do not share functional or sequence homology. It is speculated that this unique protein structure may enable better binding of hydrophobic substrates such as steroids.[3]
Although the of KSI is notably hydrophobic, it contains several hydrophilic residues believed to be important to the enzymatic function of the protein. The hydrophobic active site of KSI contains an aspartate residue at position 99 and a tyrosine residue at position 14 (according to the numbering for the Commamonas tetosteroni protein, which will be used throughout) that are capable of binding the 3-position carbonyl of the steroid. Additionally, the active site contains an aspartate residue at position 38 that is participates in the catalytic activity of KSI.[1]
Enzyme Mechanism
The general mechanism of the proposed reaction of ketosteroid isomerase involves the breaking of a C-H bond adjacent to a carbonyl. This is typically regarded as a difficult reaction due to instability of the intermediate; however it is observed in a number of enzyme-mediated biological reactions.[1] In line with other biological reactions, the mechanism of KSI involves the abstraction the β-hyrdogen from the 4-position carbon resulting in the formation of an enol intermediate, which is followed by reketonization.[1],[3] Structural and kinetic studies suggest that Asp38 (numbering is that of the TI varient of KSI) serves as a general base in this reaction as shown below.
Tyr14 and Asp99 are believed hydrogen bond to the O-3 carbonyl of the substrate steroid and stabilize reaction intermediates. Tyr14 is also believed to participate in a low barrier hydrogen bond with the 3-postition oxygen of the steroid, thereby facilitating the abstraction of the β-hydrogen at the 4-position.
Related Proteins
Available Structures
References
- ↑ 1.0 1.1 1.2 1.3 Pollack RM. Enzymatic mechanisms for catalysis of enolization: ketosteroid isomerase. Bioorg Chem. 2004 Oct;32(5):341-53. PMID:15381400 doi:10.1016/j.bioorg.2004.06.005
- ↑ 2.0 2.1 Smith SB, Richards JW, Benisek WF. The purification and characterization of delta 5-3-ketosteroid isomerase from Pseudomonas putida, a cysteine-containing isomerase. J Biol Chem. 1980 Apr 10;255(7):2678-84. PMID:7358699
- ↑ 3.0 3.1 3.2 3.3 3.4 Ha NC, Choi G, Choi KY, Oh BH. Structure and enzymology of Delta5-3-ketosteroid isomerase. Curr Opin Struct Biol. 2001 Dec;11(6):674-8. PMID:11751047
- ↑ 4.0 4.1 Wu ZR, Ebrahimian S, Zawrotny ME, Thornburg LD, Perez-Alvarado GC, Brothers P, Pollack RM, Summers MF. Solution structure of 3-oxo-delta5-steroid isomerase. Science. 1997 Apr 18;276(5311):415-8. PMID:9103200
- ↑ Cho HS, Choi G, Choi KY, Oh BH. Crystal structure and enzyme mechanism of Delta 5-3-ketosteroid isomerase from Pseudomonas testosteroni. Biochemistry. 1998 Jun 9;37(23):8325-30. PMID:9622484 doi:10.1021/bi9801614
Proteopedia Page Contributors and Editors (what is this?)
Laura M. Haynes, Michal Harel, Joel L. Sussman, Alexander Berchansky