This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Mary Ball/AFP
From Proteopedia
(→WINTER FLOUNDER ANTIFREEZE PROTEIN) |
(→WINTER FLOUNDER ANTIFREEZE PROTEIN) |
||
| Line 15: | Line 15: | ||
<scene name='User:Mary_Ball/AFP/Amino_acids/1'>See the Threonine Residues</scene> | <scene name='User:Mary_Ball/AFP/Amino_acids/1'>See the Threonine Residues</scene> | ||
| - | <scene name='User:Mary_Ball/Sandbox/Wfb/1'>WFB</scene> | + | <scene name='User:Mary_Ball/Sandbox/Wfb/1'>Go back to the WFB Alpha Helix</scene> |
| - | + | <scene name='User:Mary_Ball/AFP/Threonine_residues/2'>Scene with Threonine Residues Labelled</scene> | |
Baardsnes, et al (1999) created mutations that reduced the protein's ice-binding ability. They also compared the sequences of five different type I AFP molecules. They concluded that the "face" with the repeated Threonines promotes binding to ice crystals. | Baardsnes, et al (1999) created mutations that reduced the protein's ice-binding ability. They also compared the sequences of five different type I AFP molecules. They concluded that the "face" with the repeated Threonines promotes binding to ice crystals. | ||
Revision as of 15:05, 11 June 2010
| |||||||||
| 1wfb, resolution 1.50Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Non-Standard Residues: | |||||||||
| Related: | 1wfa | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, PDBsum, RCSB | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Contents |
WINTER FLOUNDER ANTIFREEZE PROTEIN
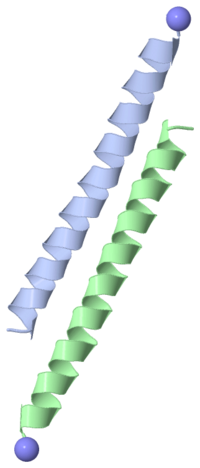
Type I antifreeze proteins (AFPs) are grouped together according to similar structure. The one shown on the right is from the winter flounder. This AFP consists of a chain of 37 amino acids that forms a single alpha-helix. Four threonines are evenly spaced in the chain, such that there are three 11-amino-acid repeats. In the single alpha-helix that forms, the threonines all lie on one "face" of the helix.
Baardsnes, et al (1999) created mutations that reduced the protein's ice-binding ability. They also compared the sequences of five different type I AFP molecules. They concluded that the "face" with the repeated Threonines promotes binding to ice crystals.
REFERENCE
New ice-binding face for type I antifreeze protein., Baardsnes J, Kondejewski LH, Hodges RS, Chao H, Kay C, Davies PL, FEBS Lett. 1999 Dec 10;463(1-2):87-91. PMID:10601644
ABOUT THIS STRUCTURE
1WFB is a 2 chains structure with sequences from Pseudopleuronectes americanus. The December 2009 RCSB PDB Molecule of the Month feature on Antifreeze Proteins by David Goodsell is 10.2210/rcsb_pdb/mom_2009_12. Full crystallographic information is available from OCA.
REFERENCE
- Sicheri F, Yang DS. Ice-binding structure and mechanism of an antifreeze protein from winter flounder. Nature. 1995 Jun 1;375(6530):427-31. PMID:7760940 doi:http://dx.doi.org/10.1038/375427a0
Page seeded by OCA on Thu Jan 21 08:56:47 2010
WINTER FLOUNDER ANTIFREEZE PROTEIN ISOFORM HPLC6 AT-180 DEGREES C
Antifreeze proteins provide fish with protection against the freezing effect of polar environments by binding to ice surfaces and inhibiting growth of ice crystals. We present the X-ray crystal structure at 1.5 A resolution of a lone alpha-helical antifreeze protein from winter flounder, which provides a detailed look at its ice-binding features. These consist of four repeated ice-binding motifs, the side chains of which are inherently rigid or restrained by pair-wise side-chain interactions to form a flat binding surface. Elaborate amino- and carboxy-terminal cap structures are also present, which explain the protein's rich alpha-helical content in solution. We propose an ice-binding model that accounts for the binding specificity of the antifreeze protein along the <0112> axes of the (2021) ice planes.
Ice-binding structure and mechanism of an antifreeze protein from winter flounder., Sicheri F, Yang DS, Nature. 1995 Jun 1;375(6530):427-31. PMID:7760940
From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.