This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
2gox
From Proteopedia
(New page: 200px<br /> <applet load="2gox" size="450" color="white" frame="true" align="right" spinBox="true" caption="2gox, resolution 2.200Å" /> '''Crystal structure ...) |
|||
| Line 1: | Line 1: | ||
| - | [[Image:2gox. | + | [[Image:2gox.jpg|left|200px]]<br /><applet load="2gox" size="350" color="white" frame="true" align="right" spinBox="true" |
| - | <applet load="2gox" size=" | + | |
caption="2gox, resolution 2.200Å" /> | caption="2gox, resolution 2.200Å" /> | ||
'''Crystal structure of Efb-C / C3d Complex'''<br /> | '''Crystal structure of Efb-C / C3d Complex'''<br /> | ||
| Line 6: | Line 5: | ||
==Overview== | ==Overview== | ||
To provide insight into bacterial suppression of complement-mediated, immunity, we present here structures of a bacterial complement inhibitory, protein, both free and bound to its complement target. The 1.25-A, structure of the complement component C3-inhibitory domain of, Staphylococcus aureus extracellular fibrinogen-binding protein (Efb-C), demonstrated a helical motif involved in complement regulation, whereas, the 2.2-A structure of Efb-C bound to the C3d domain of human C3 allowed, insight into the recognition of complement proteins by invading pathogens., Our structure-function studies provided evidence for a previously, unrecognized mode of complement inhibition whereby Efb-C binds to native, C3 and alters the solution conformation of C3 in a way that renders it, unable to participate in successful 'downstream' activation of the, complement response. | To provide insight into bacterial suppression of complement-mediated, immunity, we present here structures of a bacterial complement inhibitory, protein, both free and bound to its complement target. The 1.25-A, structure of the complement component C3-inhibitory domain of, Staphylococcus aureus extracellular fibrinogen-binding protein (Efb-C), demonstrated a helical motif involved in complement regulation, whereas, the 2.2-A structure of Efb-C bound to the C3d domain of human C3 allowed, insight into the recognition of complement proteins by invading pathogens., Our structure-function studies provided evidence for a previously, unrecognized mode of complement inhibition whereby Efb-C binds to native, C3 and alters the solution conformation of C3 in a way that renders it, unable to participate in successful 'downstream' activation of the, complement response. | ||
| - | |||
| - | ==Disease== | ||
| - | Known diseases associated with this structure: C3 deficiency OMIM:[[http://www.ncbi.nlm.nih.gov/entrez/dispomim.cgi?id=120700 120700]], Macular degeneration, age-related, 9 OMIM:[[http://www.ncbi.nlm.nih.gov/entrez/dispomim.cgi?id=120700 120700]] | ||
==About this Structure== | ==About this Structure== | ||
| - | 2GOX is a [http://en.wikipedia.org/wiki/Protein_complex Protein complex] structure of sequences from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens] and [http://en.wikipedia.org/wiki/Staphylococcus_aureus Staphylococcus aureus]. Full crystallographic information is available from [http:// | + | 2GOX is a [http://en.wikipedia.org/wiki/Protein_complex Protein complex] structure of sequences from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens] and [http://en.wikipedia.org/wiki/Staphylococcus_aureus Staphylococcus aureus]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2GOX OCA]. |
==Reference== | ==Reference== | ||
| Line 23: | Line 19: | ||
[[Category: protein-protein complex]] | [[Category: protein-protein complex]] | ||
| - | ''Page seeded by [http:// | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Wed Jan 23 13:31:53 2008'' |
Revision as of 11:31, 23 January 2008
|
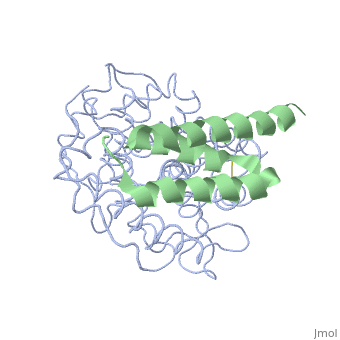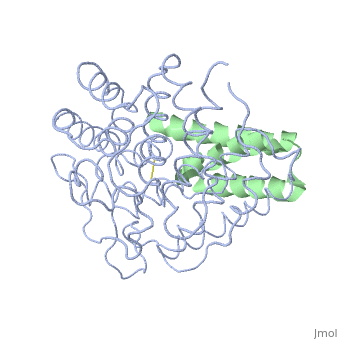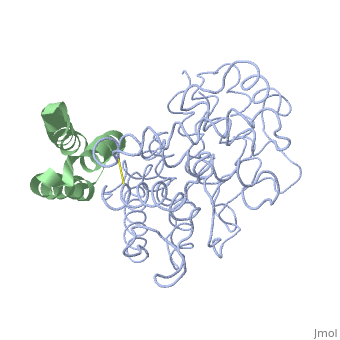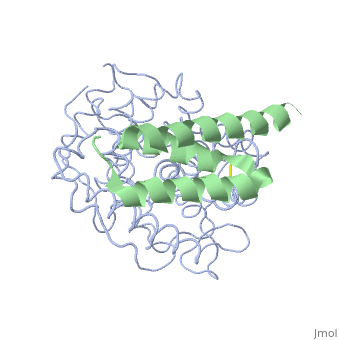
Crystal structure of Efb-C / C3d Complex
Overview
To provide insight into bacterial suppression of complement-mediated, immunity, we present here structures of a bacterial complement inhibitory, protein, both free and bound to its complement target. The 1.25-A, structure of the complement component C3-inhibitory domain of, Staphylococcus aureus extracellular fibrinogen-binding protein (Efb-C), demonstrated a helical motif involved in complement regulation, whereas, the 2.2-A structure of Efb-C bound to the C3d domain of human C3 allowed, insight into the recognition of complement proteins by invading pathogens., Our structure-function studies provided evidence for a previously, unrecognized mode of complement inhibition whereby Efb-C binds to native, C3 and alters the solution conformation of C3 in a way that renders it, unable to participate in successful 'downstream' activation of the, complement response.
About this Structure
2GOX is a Protein complex structure of sequences from Homo sapiens and Staphylococcus aureus. Full crystallographic information is available from OCA.
Reference
A structural basis for complement inhibition by Staphylococcus aureus., Hammel M, Sfyroera G, Ricklin D, Magotti P, Lambris JD, Geisbrecht BV, Nat Immunol. 2007 Apr;8(4):430-437. Epub 2007 Mar 11. PMID:17351618
Page seeded by OCA on Wed Jan 23 13:31:53 2008