Group:MUZIC:Myostatin
From Proteopedia

| Line 8: | Line 8: | ||
== Regulation of Myostatin == | == Regulation of Myostatin == | ||
| - | + | Myostatin is initially formed as a precursor protein which undergoes two proteolytic processing events in order to generate the biologically active molecule. First the N-terminal signal sequence is removed, a second generates the C-terminal fragment, which possesses receptor-binding activity <ref>PMID:9139826</ref>. | |
| - | The N-terminal fragment | + | The N-terminal fragment after proteolytic processing has been referred to as the propeptide. One mechanism for activating myostatin latency appears to be proteolytic cleavage of the propeptide <ref>PMID: 14671324 </ref>. In addition, follistatin has been known to be capable of binding and inhibiting the activity of the myostatin C-terminal dimer. <ref>PMID: 2106159 </ref>. |
[[Image:Myo1.gif]] | [[Image:Myo1.gif]] | ||
| + | (Lee,2004) | ||
| + | |||
Revision as of 15:19, 4 July 2011
|
Introduction
Myostatin (previously called GDF-8) was originally identified in a screen for novel mammalian members of the transforming growth factor-ß (TGF-ß) superfamily of growth and differentiation factors.The phenotype of myostatin knock-out mice suggested that myostatin functions as a negative regulator of muscle growth, and it was on this basis that myostatin was given its name [1]. For these reasons, inhibitors targeting myostatin has been regarded as potential drugs in the treatment of muscle-wasting disorders such as muscular dystrophy [2].
Regulation of Myostatin
Myostatin is initially formed as a precursor protein which undergoes two proteolytic processing events in order to generate the biologically active molecule. First the N-terminal signal sequence is removed, a second generates the C-terminal fragment, which possesses receptor-binding activity [3].
The N-terminal fragment after proteolytic processing has been referred to as the propeptide. One mechanism for activating myostatin latency appears to be proteolytic cleavage of the propeptide [4]. In addition, follistatin has been known to be capable of binding and inhibiting the activity of the myostatin C-terminal dimer. [5].
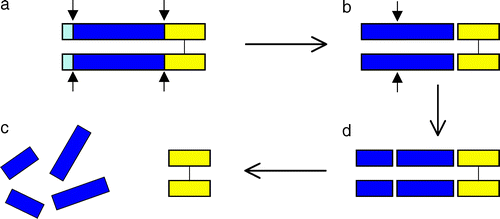 (Lee,2004)
(Lee,2004)
References
- ↑ McPherron AC, Lawler AM, Lee SJ. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature. 1997 May 1;387(6628):83-90. PMID:9139826 doi:10.1038/387083a0
- ↑ Bradley L, Yaworsky PJ, Walsh FS. Myostatin as a therapeutic target for musculoskeletal disease. Cell Mol Life Sci. 2008 Jul;65(14):2119-24. PMID:18425412 doi:10.1007/s00018-008-8077-3
- ↑ McPherron AC, Lawler AM, Lee SJ. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature. 1997 May 1;387(6628):83-90. PMID:9139826 doi:10.1038/387083a0
- ↑ Wolfman NM, McPherron AC, Pappano WN, Davies MV, Song K, Tomkinson KN, Wright JF, Zhao L, Sebald SM, Greenspan DS, Lee SJ. Activation of latent myostatin by the BMP-1/tolloid family of metalloproteinases. Proc Natl Acad Sci U S A. 2003 Dec 23;100(26):15842-6. Epub 2003 Dec 11. PMID:14671324 doi:10.1073/pnas.2534946100
- ↑ Nakamura T, Takio K, Eto Y, Shibai H, Titani K, Sugino H. Activin-binding protein from rat ovary is follistatin. Science. 1990 Feb 16;247(4944):836-8. PMID:2106159
