This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1gzx
From Proteopedia
| Line 4: | Line 4: | ||
==Overview== | ==Overview== | ||
| - | The cooperative binding of oxygen by haemoglobin results from restraints | + | The cooperative binding of oxygen by haemoglobin results from restraints on ligand binding in the T state. The unfavourable interactions made by the ligands at the haems destabilise the T state and favour the high affinity R state. The T <==> R equilibrium leads, in the presence of a ligand, to a rapid increase in the R state population and therefore generates cooperative binding. There is now considerable understanding of this phenomenon, but the interactions that reduce ligand affinity in the T state have not yet been fully explored, owing to the difficulties in preparing T state haemoglobin crystals in which all the subunits are oxygenated. A protocol has been developed to oxygenate deoxy T state adult human haemoglobin (HbA) crystals in air at 4 C at all four haems without significant loss of crystalline order. The X-ray crystal structure, determined to 2.1 A spacing, shows significant changes in the alpha and beta haem pockets as well as changes at the alpha(1)beta(2) interface in the direction of the R quaternary structure. Most of the shifts and deviations from deoxy T state HbA are similar to, but larger than, those previously observed in the T state met and other partially liganded T state forms. They provide clear evidence of haem-haem interaction in the T state. |
==Disease== | ==Disease== | ||
| Line 28: | Line 28: | ||
[[Category: transport]] | [[Category: transport]] | ||
| - | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Feb 21 12:55:50 2008'' |
Revision as of 10:55, 21 February 2008
|
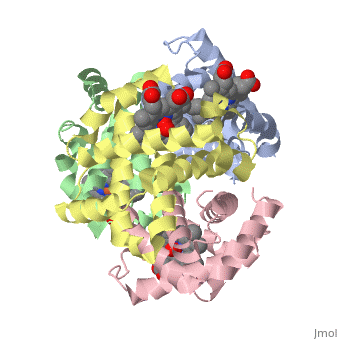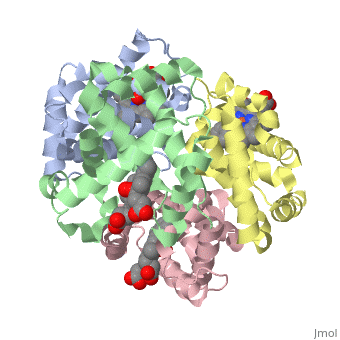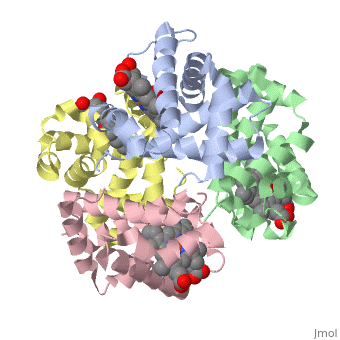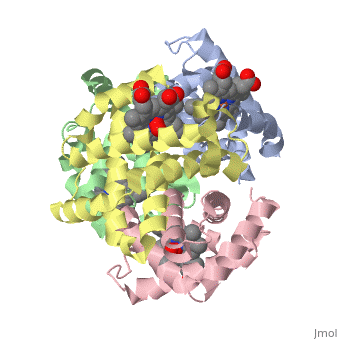
OXY T STATE HAEMOGLOBIN: OXYGEN BOUND AT ALL FOUR HAEMS
Contents |
Overview
The cooperative binding of oxygen by haemoglobin results from restraints on ligand binding in the T state. The unfavourable interactions made by the ligands at the haems destabilise the T state and favour the high affinity R state. The T <==> R equilibrium leads, in the presence of a ligand, to a rapid increase in the R state population and therefore generates cooperative binding. There is now considerable understanding of this phenomenon, but the interactions that reduce ligand affinity in the T state have not yet been fully explored, owing to the difficulties in preparing T state haemoglobin crystals in which all the subunits are oxygenated. A protocol has been developed to oxygenate deoxy T state adult human haemoglobin (HbA) crystals in air at 4 C at all four haems without significant loss of crystalline order. The X-ray crystal structure, determined to 2.1 A spacing, shows significant changes in the alpha and beta haem pockets as well as changes at the alpha(1)beta(2) interface in the direction of the R quaternary structure. Most of the shifts and deviations from deoxy T state HbA are similar to, but larger than, those previously observed in the T state met and other partially liganded T state forms. They provide clear evidence of haem-haem interaction in the T state.
Disease
Known diseases associated with this structure: Erythremias, alpha- OMIM:[141800], Erythremias, beta- OMIM:[141900], Erythrocytosis OMIM:[141850], HPFH, deletion type OMIM:[141900], Heinz body anemia OMIM:[141850], Heinz body anemias, alpha- OMIM:[141800], Heinz body anemias, beta- OMIM:[141900], Hemoglobin H disease OMIM:[141850], Hypochromic microcytic anemia OMIM:[141850], Methemoglobinemias, alpha- OMIM:[141800], Methemoglobinemias, beta- OMIM:[141900], Sickle cell anemia OMIM:[141900], Thalassemia, alpha- OMIM:[141850], Thalassemia-beta, dominant inclusion-body OMIM:[141900], Thalassemias, alpha- OMIM:[141800], Thalassemias, beta- OMIM:[141900]
About this Structure
1GZX is a Protein complex structure of sequences from Homo sapiens with and as ligands. Known structural/functional Site: . Full crystallographic information is available from OCA.
Reference
Crystal structure of T state haemoglobin with oxygen bound at all four haems., Paoli M, Liddington R, Tame J, Wilkinson A, Dodson G, J Mol Biol. 1996 Mar 8;256(4):775-92. PMID:8642597
Page seeded by OCA on Thu Feb 21 12:55:50 2008
Categories: Homo sapiens | Protein complex | Dodson, G. | Liddington, R. | Paoli, M. | Tame, J. | Wilkinson, A. | HEM | OXY | Cooperativity | Haem protein | Oxygen binding | Transport