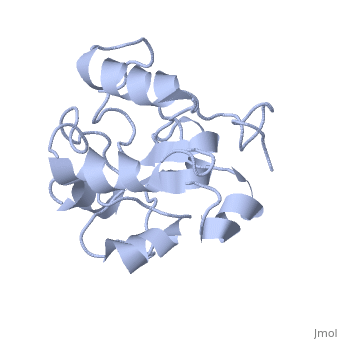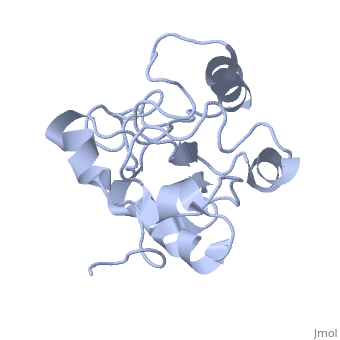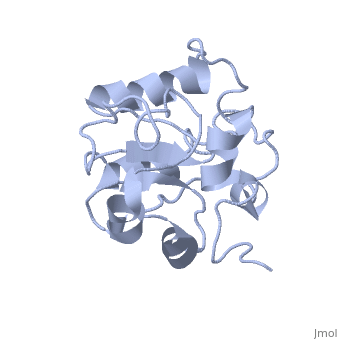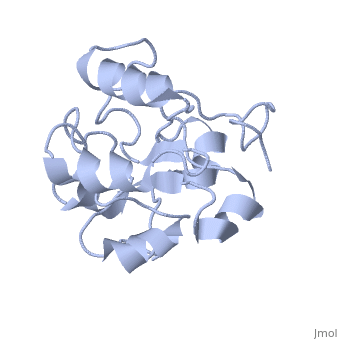This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1hzm
From Proteopedia
| Line 4: | Line 4: | ||
==Overview== | ==Overview== | ||
| - | MAP kinases (MAPKs), which control mitogenic signal transduction in all | + | MAP kinases (MAPKs), which control mitogenic signal transduction in all eukaryotic organisms, are inactivated by dual specificity MAPK phosphatases (MKPs). MKP-3, a prototypical MKP, achieves substrate specificity through its N-terminal domain binding to the MAPK ERK2, resulting in the activation of its C-terminal phosphatase domain. The solution structure and biochemical analysis of the ERK2 binding (EB) domain of MKP-3 show that regions that are essential for ERK2 binding partly overlap with its sites that interact with the C-terminal catalytic domain, and that these interactions are functionally coupled to the active site residues of MKP-3. Our findings suggest a novel mechanism by which the EB domain binding to ERK2 is transduced to cause a conformational change of the C-terminal catalytic domain, resulting in the enzymatic activation of MKP-3. |
==About this Structure== | ==About this Structure== | ||
| Line 14: | Line 14: | ||
[[Category: Single protein]] | [[Category: Single protein]] | ||
[[Category: Farooq, A.]] | [[Category: Farooq, A.]] | ||
| - | [[Category: Zhou, M | + | [[Category: Zhou, M M.]] |
[[Category: hydrolase]] | [[Category: hydrolase]] | ||
| - | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Feb 21 13:06:26 2008'' |
Revision as of 11:06, 21 February 2008
|
STRUCTURE OF ERK2 BINDING DOMAIN OF MAPK PHOSPHATASE MKP-3: STRUCTURAL INSIGHTS INTO MKP-3 ACTIVATION BY ERK2
Overview
MAP kinases (MAPKs), which control mitogenic signal transduction in all eukaryotic organisms, are inactivated by dual specificity MAPK phosphatases (MKPs). MKP-3, a prototypical MKP, achieves substrate specificity through its N-terminal domain binding to the MAPK ERK2, resulting in the activation of its C-terminal phosphatase domain. The solution structure and biochemical analysis of the ERK2 binding (EB) domain of MKP-3 show that regions that are essential for ERK2 binding partly overlap with its sites that interact with the C-terminal catalytic domain, and that these interactions are functionally coupled to the active site residues of MKP-3. Our findings suggest a novel mechanism by which the EB domain binding to ERK2 is transduced to cause a conformational change of the C-terminal catalytic domain, resulting in the enzymatic activation of MKP-3.
About this Structure
1HZM is a Single protein structure of sequence from Homo sapiens. Full crystallographic information is available from OCA.
Reference
Solution structure of ERK2 binding domain of MAPK phosphatase MKP-3: structural insights into MKP-3 activation by ERK2., Farooq A, Chaturvedi G, Mujtaba S, Plotnikova O, Zeng L, Dhalluin C, Ashton R, Zhou MM, Mol Cell. 2001 Feb;7(2):387-99. PMID:11239467
Page seeded by OCA on Thu Feb 21 13:06:26 2008