This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1iat
From Proteopedia
(New page: 200px<br /> <applet load="1iat" size="450" color="white" frame="true" align="right" spinBox="true" caption="1iat, resolution 1.62Å" /> '''CRYSTAL STRUCTURE O...) |
|||
| Line 1: | Line 1: | ||
| - | [[Image:1iat.gif|left|200px]]<br /> | + | [[Image:1iat.gif|left|200px]]<br /><applet load="1iat" size="350" color="white" frame="true" align="right" spinBox="true" |
| - | <applet load="1iat" size=" | + | |
caption="1iat, resolution 1.62Å" /> | caption="1iat, resolution 1.62Å" /> | ||
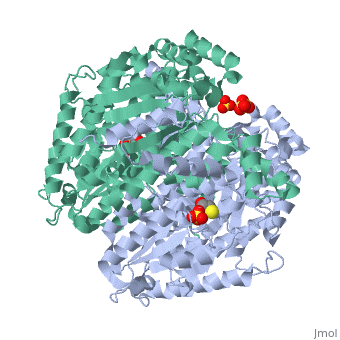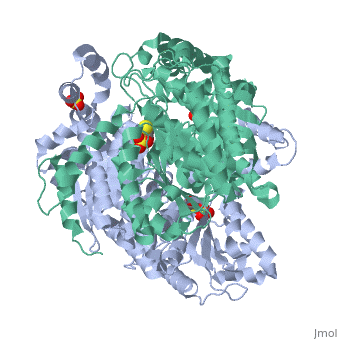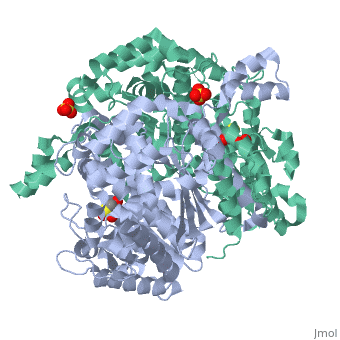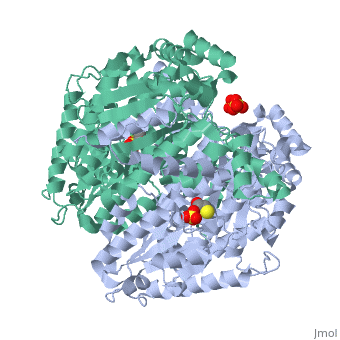
'''CRYSTAL STRUCTURE OF HUMAN PHOSPHOGLUCOSE ISOMERASE/NEUROLEUKIN/AUTOCRINE MOTILITY FACTOR/MATURATION FACTOR'''<br /> | '''CRYSTAL STRUCTURE OF HUMAN PHOSPHOGLUCOSE ISOMERASE/NEUROLEUKIN/AUTOCRINE MOTILITY FACTOR/MATURATION FACTOR'''<br /> | ||
==Overview== | ==Overview== | ||
| - | Phosphoglucose isomerase (PGI) is a multifunctional protein, which, inside | + | Phosphoglucose isomerase (PGI) is a multifunctional protein, which, inside the cell, functions as a housekeeping enzyme of glycolysis and gluconeogenesis and, outside the cell, exerts wholly unrelated cytokine properties. We have determined the structure of human PGI to a resolution of 1.6 A using X-ray crystallography. The structure is highly similar to other PGIs, especially the architecture of the active site. Fortuitous binding of a sulphate molecule from the crystallisation solution has facilitated an accurate description of the substrate phosphate-binding site. Comparison with both native and inhibitor-bound rabbit PGI structures shows that two loops move closer to the active site upon binding inhibitor. Interestingly, the human structure most closely resembles the inhibitor-bound structure, suggesting that binding of the phosphate moiety of the substrate may trigger this conformational change. We suggest a new mechanism for catalysis that uses Glu357 as the base catalyst for the isomerase reaction rather than His388 as proposed previously. The human PGI structure has also provided a detailed framework with which to map mutations associated with non-spherocytic haemolytic anaemia. |
==Disease== | ==Disease== | ||
| Line 11: | Line 10: | ||
==About this Structure== | ==About this Structure== | ||
| - | 1IAT is a [http://en.wikipedia.org/wiki/Single_protein Single protein] structure of sequence from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens] with SO4 and BME as [http://en.wikipedia.org/wiki/ligands ligands]. Active as [http://en.wikipedia.org/wiki/Glucose-6-phosphate_isomerase Glucose-6-phosphate isomerase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=5.3.1.9 5.3.1.9] Full crystallographic information is available from [http:// | + | 1IAT is a [http://en.wikipedia.org/wiki/Single_protein Single protein] structure of sequence from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens] with <scene name='pdbligand=SO4:'>SO4</scene> and <scene name='pdbligand=BME:'>BME</scene> as [http://en.wikipedia.org/wiki/ligands ligands]. Active as [http://en.wikipedia.org/wiki/Glucose-6-phosphate_isomerase Glucose-6-phosphate isomerase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=5.3.1.9 5.3.1.9] Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1IAT OCA]. |
==Reference== | ==Reference== | ||
| Line 23: | Line 22: | ||
[[Category: Muirhead, H.]] | [[Category: Muirhead, H.]] | ||
[[Category: Pearce, J.]] | [[Category: Pearce, J.]] | ||
| - | [[Category: Read, J | + | [[Category: Read, J A.]] |
[[Category: BME]] | [[Category: BME]] | ||
[[Category: SO4]] | [[Category: SO4]] | ||
| Line 30: | Line 29: | ||
[[Category: two alpha/beta domains]] | [[Category: two alpha/beta domains]] | ||
| - | ''Page seeded by [http:// | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Feb 21 13:09:49 2008'' |
Revision as of 11:09, 21 February 2008
|
CRYSTAL STRUCTURE OF HUMAN PHOSPHOGLUCOSE ISOMERASE/NEUROLEUKIN/AUTOCRINE MOTILITY FACTOR/MATURATION FACTOR
Contents |
Overview
Phosphoglucose isomerase (PGI) is a multifunctional protein, which, inside the cell, functions as a housekeeping enzyme of glycolysis and gluconeogenesis and, outside the cell, exerts wholly unrelated cytokine properties. We have determined the structure of human PGI to a resolution of 1.6 A using X-ray crystallography. The structure is highly similar to other PGIs, especially the architecture of the active site. Fortuitous binding of a sulphate molecule from the crystallisation solution has facilitated an accurate description of the substrate phosphate-binding site. Comparison with both native and inhibitor-bound rabbit PGI structures shows that two loops move closer to the active site upon binding inhibitor. Interestingly, the human structure most closely resembles the inhibitor-bound structure, suggesting that binding of the phosphate moiety of the substrate may trigger this conformational change. We suggest a new mechanism for catalysis that uses Glu357 as the base catalyst for the isomerase reaction rather than His388 as proposed previously. The human PGI structure has also provided a detailed framework with which to map mutations associated with non-spherocytic haemolytic anaemia.
Disease
Known diseases associated with this structure: Hemolytic anemia due to glucosephosphate isomerase deficiency OMIM:[172400], Hydrops fetalis, one form OMIM:[172400]
About this Structure
1IAT is a Single protein structure of sequence from Homo sapiens with and as ligands. Active as Glucose-6-phosphate isomerase, with EC number 5.3.1.9 Full crystallographic information is available from OCA.
Reference
The crystal structure of human phosphoglucose isomerase at 1.6 A resolution: implications for catalytic mechanism, cytokine activity and haemolytic anaemia., Read J, Pearce J, Li X, Muirhead H, Chirgwin J, Davies C, J Mol Biol. 2001 Jun 1;309(2):447-63. PMID:11371164
Page seeded by OCA on Thu Feb 21 13:09:49 2008