This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1ktz
From Proteopedia
(New page: 200px<br /> <applet load="1ktz" size="450" color="white" frame="true" align="right" spinBox="true" caption="1ktz, resolution 2.15Å" /> '''Crystal Structure o...) |
|||
| Line 1: | Line 1: | ||
| - | [[Image:1ktz.gif|left|200px]]<br /> | + | [[Image:1ktz.gif|left|200px]]<br /><applet load="1ktz" size="350" color="white" frame="true" align="right" spinBox="true" |
| - | <applet load="1ktz" size=" | + | |
caption="1ktz, resolution 2.15Å" /> | caption="1ktz, resolution 2.15Å" /> | ||
'''Crystal Structure of the Human TGF-beta Type II Receptor Extracellular Domain in Complex with TGF-beta3'''<br /> | '''Crystal Structure of the Human TGF-beta Type II Receptor Extracellular Domain in Complex with TGF-beta3'''<br /> | ||
==Overview== | ==Overview== | ||
| - | Transforming growth factor-beta (TGF-beta) is the prototype of a large | + | Transforming growth factor-beta (TGF-beta) is the prototype of a large family of structurally related cytokines that play key roles in maintaining cellular homeostasis by signaling through two classes of functionally distinct Ser/Thr kinase receptors, designated as type I and type II. TGF-beta initiates receptor assembly by binding with high affinity to the type II receptor. Here, we present the 2.15 A crystal structure of the extracellular ligand-binding domain of the human TGF-beta type II receptor (ecTbetaR2) in complex with human TGF-beta3. ecTbetaR2 interacts with homodimeric TGF-beta3 by binding identical finger segments at opposite ends of the growth factor. Relative to the canonical 'closed' conformation previously observed in ligand structures across the superfamily, ecTbetaR2-bound TGF-beta3 shows an altered arrangement of its monomeric subunits, designated the 'open' conformation. The mode of TGF-beta3 binding shown by ecTbetaR2 is compatible with both ligand conformations. This, in addition to the predicted mode for TGF-beta binding to the type I receptor ectodomain (ecTbetaR1), suggests an assembly mechanism in which ecTbetaR1 and ecTbetaR2 bind at adjacent positions on the ligand surface and directly contact each other via protein--protein interactions. |
==Disease== | ==Disease== | ||
| Line 11: | Line 10: | ||
==About this Structure== | ==About this Structure== | ||
| - | 1KTZ is a [http://en.wikipedia.org/wiki/Protein_complex Protein complex] structure of sequences from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens]. Active as [http://en.wikipedia.org/wiki/Non-specific_serine/threonine_protein_kinase Non-specific serine/threonine protein kinase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=2.7.11.1 2.7.11.1] Full crystallographic information is available from [http:// | + | 1KTZ is a [http://en.wikipedia.org/wiki/Protein_complex Protein complex] structure of sequences from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens]. Active as [http://en.wikipedia.org/wiki/Non-specific_serine/threonine_protein_kinase Non-specific serine/threonine protein kinase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=2.7.11.1 2.7.11.1] Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1KTZ OCA]. |
==Reference== | ==Reference== | ||
| Line 19: | Line 18: | ||
[[Category: Protein complex]] | [[Category: Protein complex]] | ||
[[Category: Deep, S.]] | [[Category: Deep, S.]] | ||
| - | [[Category: Hart, P | + | [[Category: Hart, P J.]] |
| - | [[Category: Hinck, A | + | [[Category: Hinck, A P.]] |
| - | [[Category: Hinck, C | + | [[Category: Hinck, C S.]] |
[[Category: Shu, Z.]] | [[Category: Shu, Z.]] | ||
| - | [[Category: Taylor, A | + | [[Category: Taylor, A B.]] |
[[Category: cytokine-receptor complex]] | [[Category: cytokine-receptor complex]] | ||
| - | ''Page seeded by [http:// | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Feb 21 13:37:53 2008'' |
Revision as of 11:37, 21 February 2008
|
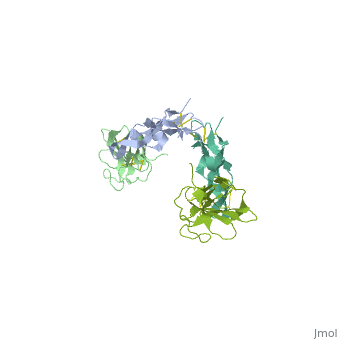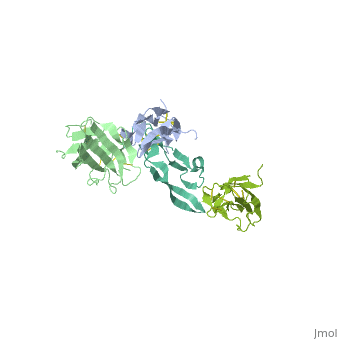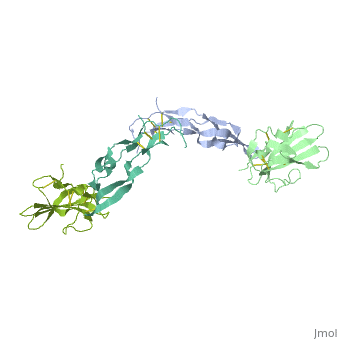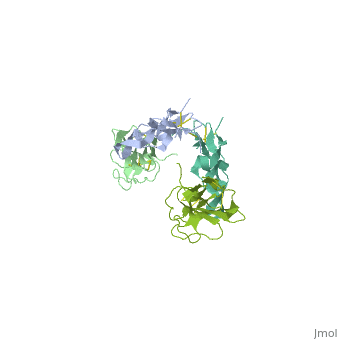
Crystal Structure of the Human TGF-beta Type II Receptor Extracellular Domain in Complex with TGF-beta3
Contents |
Overview
Transforming growth factor-beta (TGF-beta) is the prototype of a large family of structurally related cytokines that play key roles in maintaining cellular homeostasis by signaling through two classes of functionally distinct Ser/Thr kinase receptors, designated as type I and type II. TGF-beta initiates receptor assembly by binding with high affinity to the type II receptor. Here, we present the 2.15 A crystal structure of the extracellular ligand-binding domain of the human TGF-beta type II receptor (ecTbetaR2) in complex with human TGF-beta3. ecTbetaR2 interacts with homodimeric TGF-beta3 by binding identical finger segments at opposite ends of the growth factor. Relative to the canonical 'closed' conformation previously observed in ligand structures across the superfamily, ecTbetaR2-bound TGF-beta3 shows an altered arrangement of its monomeric subunits, designated the 'open' conformation. The mode of TGF-beta3 binding shown by ecTbetaR2 is compatible with both ligand conformations. This, in addition to the predicted mode for TGF-beta binding to the type I receptor ectodomain (ecTbetaR1), suggests an assembly mechanism in which ecTbetaR1 and ecTbetaR2 bind at adjacent positions on the ligand surface and directly contact each other via protein--protein interactions.
Disease
Known diseases associated with this structure: Aortic aneurysm, familial thoracic 3 OMIM:[190182], Arrhythmogenic right ventricular dysplasia 1 OMIM:[190230], Colon cancer OMIM:[190182], Colorectal cancer, hereditary nonpolyposis, type 6 OMIM:[190182], Esophageal cancer OMIM:[190182], Loeys-Dietz syndrome OMIM:[190182], Marfan syndrome, type II OMIM:[190182]
About this Structure
1KTZ is a Protein complex structure of sequences from Homo sapiens. Active as Non-specific serine/threonine protein kinase, with EC number 2.7.11.1 Full crystallographic information is available from OCA.
Reference
Crystal structure of the human TbetaR2 ectodomain--TGF-beta3 complex., Hart PJ, Deep S, Taylor AB, Shu Z, Hinck CS, Hinck AP, Nat Struct Biol. 2002 Mar;9(3):203-8. PMID:11850637
Page seeded by OCA on Thu Feb 21 13:37:53 2008