This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Fragment-Based Drug Discovery
From Proteopedia
(Difference between revisions)
| Line 27: | Line 27: | ||
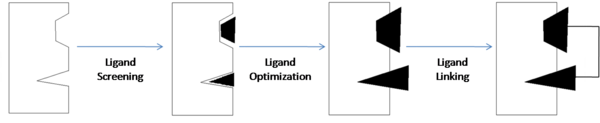
Three ligands with moderate affinity for Bcl-xl were analyzed using SAR by NMR in order to develop ABT-737. The structural components that allow the ligands to bind to the protein were then linked together to form ABT-737 - the final compound with high-affinity for Bcl-xl. | Three ligands with moderate affinity for Bcl-xl were analyzed using SAR by NMR in order to develop ABT-737. The structural components that allow the ligands to bind to the protein were then linked together to form ABT-737 - the final compound with high-affinity for Bcl-xl. | ||
| - | <scene name='Sandbox_reserved_394/Compound_1/2'>Compound 1</scene> is a 4'-fluoro-biphenyl-4-carboxylic acid. SAR by NMR was used to identify the interactions that this compound forms with Bcl-xl. The fluorobiphenyl system is hydrophobic and its interactions form a <scene name='Sandbox_reserved_394/Compound_1/4'>"hydrophobic pocket"</scene> around the fluorobiphenyl system. The <scene name='Sandbox_reserved_394/Compound_1/5'>carboxyilic acid portion binds near Gly 142</scene> of Bcl-xl. The carboxylic acid is later substituted with an acyl sulfonamide (shown in | + | <scene name='Sandbox_reserved_394/Compound_1/2'>Compound 1</scene> is a 4'-fluoro-biphenyl-4-carboxylic acid. SAR by NMR was used to identify the interactions that this compound forms with Bcl-xl. The fluorobiphenyl system is hydrophobic and its interactions form a <scene name='Sandbox_reserved_394/Compound_1/4'>"hydrophobic pocket"</scene> around the fluorobiphenyl system. The <scene name='Sandbox_reserved_394/Compound_1/5'>carboxyilic acid portion binds near Gly 142</scene> of Bcl-xl. The carboxylic acid is later substituted with an acyl sulfonamide (shown in compound 2) which provides increased affinity. |
| - | <scene name='Sandbox_reserved_394/Compound_2/1'>Compound 2</scene> binds with high affinity to Bcl-xl. | + | <scene name='Sandbox_reserved_394/Compound_2/1'>Compound 2</scene> binds with high affinity to Bcl-xl. The <scene name='Sandbox_reserved_394/Compound_2/2'>acylsulfonamide portion forms a hydrogen bond with Gly 142</scene>. The substitution of this sulfonamide for the carboxylic acid from compound 1 allows compound 2 to form a much stronger bond with Bcl-xl by bringing the shared acidic proton in closer proximity to GLY 142. The binding affinity of compound 2 for Bcl-xl is greatly reduced in the presence of human serum albumin (HSA). In order to decrease HSA affinity, and therefore increase Bcl-xl affinity, SAR by NMR was used to modify compound 1 by eliminating key binding groups of compound 1 to HSA without affecting Bcl-xl affinity. |
{| class="wikitable collapsible collapsed" | {| class="wikitable collapsible collapsed" | ||
| - | ! scope="col" width="5000px" | Modifying compound | + | ! scope="col" width="5000px" | Modifying compound 2 to reduce HSA affinity |
|- | |- | ||
| - | | scope="col" width="5000px" | this | + | | scope="col" width="5000px" | Compound 2 has high affinity for Bcl-xl but has an even higher affinity for HSA. For this reason, when HSA is present, compound 2 and similar ligands are more likely to bind to HSA thereby decreasing the amount that can bind with Bcl-xl. In order to decrease the affinity for HSA while maintaining affinity for Bcl-xl, SAR by NMR was used to compare compound 2 with a <scene name='Sandbox_reserved_394/Compound_3/1'>thioethylamino-2,4-dimethylphenyl analogue</scene>, which also has high affinity for HSA. It was found that two hydrophobic portions of compound 2 had very strong hydrophobic interactions with HSA. Therefore, these portions were modified with polar substituents to decrease HSA affinity. To decrease hydrophobicity, the fluorobiphenyl system was substituted with a piperazine ring and a 2-dimethylaminoethyl group was added to the thioethylamino linkage group. |
|} | |} | ||
Revision as of 04:19, 30 October 2012
Drug Design: SAR by NMR
| |||||||||||
References
- ↑ Pandit D. LIGAND-BASED DRUG DESIGN: I. CONFORMATIONAL STUDIES OF GBR 12909 ANALOGS AS COCAINE ANTAGONISTS; II. 3D-QSAR STUDIES OF SALVINORIN A ANALOGS AS εΑΡΡΑ OPIOID AGONISTS. http://archives.njit.edu/vol01/etd/2000s/2007/njit-etd2007-051/njit-etd2007-051.pdf
- ↑ Shuker S. B., Hajduk P. J., Meadows R. P., Fesik S. W. Discovering High-Affinity Ligands for Proteins: SAR by NMR. Science; Nov 29, 1996; 274, 5292; ProQuest Central pg. 1531.
- ↑ Oltersdorf T., Elmore S. W., Shoemaker A. R. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Vol 435|2 June 2005|doi:10.1038/nature03579