This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Fragment-Based Drug Discovery
From Proteopedia
(Difference between revisions)
| Line 9: | Line 9: | ||
---- | ---- | ||
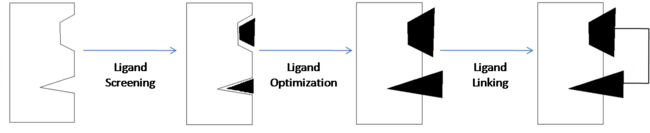
| - | '''Fragment-based drug discovery''' (FBDD) is a method of discovering new compounds by utilizing fragments that have some degree of binding affinity for a drug target, optimizing those fragments so as to increase their binding affinity, then linking them together to form a lead compound that has high affinity and selectivity for the drug target. Nuclear magnetic resonance (NMR) and x-ray crystallography can be used to analyze the fragments and drug targets in order to create three-dimensional images which can be used to obtain an analysis of molecular relationships. This allows developers to get a visual representation of how each fragment binds to the target | + | '''Fragment-based drug discovery''' (FBDD) is a method of discovering new compounds by utilizing fragments that have some degree of binding affinity for a drug target, optimizing those fragments so as to increase their binding affinity, then linking them together to form a lead compound that has high affinity and selectivity for the drug target. Nuclear magnetic resonance (NMR) and x-ray crystallography can be used to analyze the fragments and drug targets in order to create three-dimensional images which can be used to obtain an analysis of molecular relationships. This allows developers to get a visual representation of how each fragment binds to the target and can also be useful in identifying the individual binding sites of the target. |
[[Image:SAR by NMR Illustrated.png | thumb | center | 650px | Fragment-Based Drug Discovery (Adapted from Fig. 1)<ref name="Shuker S. B., Hajduk P. J., Meadows R. P., Fesik S. W. Discovering High-Affinity Ligands for Proteins: SAR by NMR. Science; Nov 29, 1996; 274, 5292; ProQuest Central pg. 1531.">Shuker S. B., Hajduk P. J., Meadows R. P., Fesik S. W. Discovering High-Affinity Ligands for Proteins: SAR by NMR. Science; Nov 29, 1996; 274, 5292; ProQuest Central pg. 1531.</ref>]] | [[Image:SAR by NMR Illustrated.png | thumb | center | 650px | Fragment-Based Drug Discovery (Adapted from Fig. 1)<ref name="Shuker S. B., Hajduk P. J., Meadows R. P., Fesik S. W. Discovering High-Affinity Ligands for Proteins: SAR by NMR. Science; Nov 29, 1996; 274, 5292; ProQuest Central pg. 1531.">Shuker S. B., Hajduk P. J., Meadows R. P., Fesik S. W. Discovering High-Affinity Ligands for Proteins: SAR by NMR. Science; Nov 29, 1996; 274, 5292; ProQuest Central pg. 1531.</ref>]] | ||
Revision as of 18:58, 31 October 2012
Drug Design: Fragment-Based Drug Discovery
| |||||||||||
References
- ↑ 1.0 1.1 Shuker S. B., Hajduk P. J., Meadows R. P., Fesik S. W. Discovering High-Affinity Ligands for Proteins: SAR by NMR. Science; Nov 29, 1996; 274, 5292; ProQuest Central pg. 1531.
- ↑ Oltersdorf T., Elmore S. W., Shoemaker A. R. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Vol 435|2 June 2005|doi:10.1038/nature03579
- ↑ Pandit D. LIGAND-BASED DRUG DESIGN: I. CONFORMATIONAL STUDIES OF GBR 12909 ANALOGS AS COCAINE ANTAGONISTS; II. 3D-QSAR STUDIES OF SALVINORIN A ANALOGS AS εΑΡΡΑ OPIOID AGONISTS. http://archives.njit.edu/vol01/etd/2000s/2007/njit-etd2007-051/njit-etd2007-051.pdf