This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 390
From Proteopedia
(Difference between revisions)
| Line 20: | Line 20: | ||
==COMPOUND ACTIVE SITE== | ==COMPOUND ACTIVE SITE== | ||
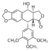
At the <scene name='Sandbox_Reserved_390/Top/12'>active site</scene> we can see how the <scene name='Sandbox_Reserved_390/Top/14'>ligand</scene> (in green) is stabilized. This molecule has approximately 36 <scene name='Sandbox_Reserved_390/Top/3'>alpha helices</scene> represented with magenta helices and approximately 40 <scene name='Sandbox_Reserved_390/Top/2'>beta sheets</scene> represented with blue arrows in the <scene name='Sandbox_Reserved_390/Top/4'>secondary structures</scene>. | At the <scene name='Sandbox_Reserved_390/Top/12'>active site</scene> we can see how the <scene name='Sandbox_Reserved_390/Top/14'>ligand</scene> (in green) is stabilized. This molecule has approximately 36 <scene name='Sandbox_Reserved_390/Top/3'>alpha helices</scene> represented with magenta helices and approximately 40 <scene name='Sandbox_Reserved_390/Top/2'>beta sheets</scene> represented with blue arrows in the <scene name='Sandbox_Reserved_390/Top/4'>secondary structures</scene>. | ||
| - | |||
| - | ==STRUCTURE-ACTIVITY RELATIONS (SAR)== | ||
| - | This interplay between the protein, the DNA, and the drug explains the structure-activity relations of etoposide derivatives and the molecular basis of drug-resistant mutations. This resistance occurs via two mechanisms: '''1)''' Decreased accumulation via increased P-glycoprotein; and '''2)''' Changes in target proteins (mutation or decreased expression of topoisomerase II or decreased apoptosis due to mutation of p53). | ||
==ETOPOSIDE RESISTANCE== | ==ETOPOSIDE RESISTANCE== | ||
| + | The interplay between the protein, the DNA, and the drug explains the structure-activity relations of etoposide derivatives and the molecular basis of drug-resistant mutations. This resistance occurs via two mechanisms: '''1)''' Decreased accumulation via increased P-glycoprotein; and '''2)''' Changes in target proteins (mutation or decreased expression of topoisomerase II or decreased apoptosis due to mutation of p53). | ||
'''1.''' Decreased accumulation via increased P-glycoprotein (a multidrug resistance): This drug resistance mechanism is characterized by decreased intracellular accumulation of drug facilitated by overexpression of the human multidrug resistance (mdrl) gene, causing overproduction of P-glycoprotein. This cell membrane protein acts as an export pump for a wide variety of unrelated foreign natural products. By maintaining lower intracellular levels of drug, fewer drugs would be available to the target, which is topoisomerase II. | '''1.''' Decreased accumulation via increased P-glycoprotein (a multidrug resistance): This drug resistance mechanism is characterized by decreased intracellular accumulation of drug facilitated by overexpression of the human multidrug resistance (mdrl) gene, causing overproduction of P-glycoprotein. This cell membrane protein acts as an export pump for a wide variety of unrelated foreign natural products. By maintaining lower intracellular levels of drug, fewer drugs would be available to the target, which is topoisomerase II. | ||
Revision as of 14:41, 12 November 2012
Human topoisomerase IIbeta in complex with DNA and etoposide (Vepesid)
| |||||||||||
References
- ↑ Kathryn L. Gilroy, Chrysoula Leontiou, Kay Padget, Jeremy H. Lakey and Caroline A. Austin* "mAMSA resistant human topoisomerase IIβ mutation G465D has reduced ATP hydrolysis activity” Oxford JournalsLife Sciences Nucleic Acids Research Volume 34, Issue 5Pp. 1597-1607. DOI: 10.1093/nar/gkl057
- ↑ Wu CC, Li TK, Farh L, Lin LY, Lin TS, Yu YJ, Yen TJ, Chiang CW, Chan NL. Structural basis of type II topoisomerase inhibition by the anticancer drug etoposide. Science. 2011 Jul 22;333(6041):459-62. PMID:21778401 doi:10.1126/science.1204117