This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Group:MUZIC:Myostatin
From Proteopedia

(→Introduction) |
(→Regulation of Myostatin) |
||
| Line 7: | Line 7: | ||
== Regulation of Myostatin == | == Regulation of Myostatin == | ||
| - | Myostatin is initially formed as a precursor protein which undergoes two proteolytic processing events in order to generate the biologically active molecule. First the N-terminal signal sequence is removed, a second generates the C-terminal fragment, which possesses receptor-binding activity <ref>PMID:9139826</ref>. | + | Myostatin is initially formed as a precursor protein which undergoes two proteolytic processing events in order to generate the biologically active molecule. First the N-terminal signal sequence is removed, a second cleavage generates the C-terminal fragment, which possesses receptor-binding activity <ref>PMID:9139826</ref>. |
| - | The N-terminal fragment after proteolytic processing has been referred to as the propeptide. One mechanism for activating myostatin latency appears to be proteolytic cleavage of the propeptide <ref>PMID: 14671324 </ref>. In addition, follistatin has been known to be capable of binding and inhibiting the activity of the myostatin C-terminal dimer. <ref>PMID: 2106159 </ref>. | + | The N-terminal fragment after proteolytic processing has been referred to as the propeptide. One mechanism for activating myostatin latency appears to be proteolytic cleavage of the propeptide <ref>PMID: 14671324 </ref>. In addition to the regulation of intracellular myostation processing,follistatin has been known to be capable of binding and inhibiting the activity of the myostatin C-terminal dimer. <ref>PMID: 2106159 </ref>. |
[[Image:Myo1.gif]] | [[Image:Myo1.gif]] | ||
(Lee,2004) | (Lee,2004) | ||
| - | |||
== Structure == | == Structure == | ||
Revision as of 12:50, 28 November 2012
|
Contents |
Introduction
Myostatin which is also known as growth and developmental factor-8(GDF-8) was originally identified in a screen for novel mammalian members of the transforming growth factor-ß (TGF-ß) superfamily of growth and differentiation factors.The phenotype of myostatin knock-out mice suggested that myostatin functions as a negative regulator of muscle growth, and it was on this basis that myostatin was given its name [1]. For these reasons, inhibitors targeting myostatin has been regarded as potential drugs in the treatment of muscle-wasting disorders such as muscular dystrophy [2].
Regulation of Myostatin
Myostatin is initially formed as a precursor protein which undergoes two proteolytic processing events in order to generate the biologically active molecule. First the N-terminal signal sequence is removed, a second cleavage generates the C-terminal fragment, which possesses receptor-binding activity [3].
The N-terminal fragment after proteolytic processing has been referred to as the propeptide. One mechanism for activating myostatin latency appears to be proteolytic cleavage of the propeptide [4]. In addition to the regulation of intracellular myostation processing,follistatin has been known to be capable of binding and inhibiting the activity of the myostatin C-terminal dimer. [5].
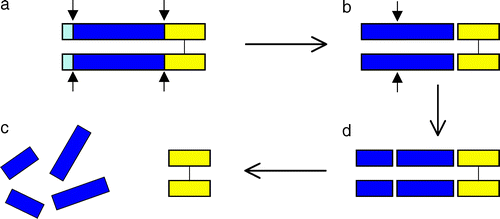 (Lee,2004)
(Lee,2004)
Structure
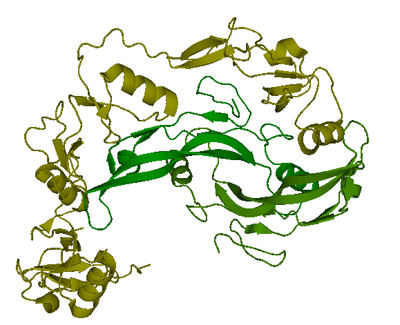
Only one structure of myostatin is currently available in Protein Data Bank(PDB). The complex of two follistatin 288 molecules bound to one myostatin dimer was resolved to 2.15 Å using X-ray crystallography and deposited in PDB. (Green:myostatin C-terminal dimer, yellow: follinstatin 288)
References
- ↑ McPherron AC, Lawler AM, Lee SJ. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature. 1997 May 1;387(6628):83-90. PMID:9139826 doi:10.1038/387083a0
- ↑ Bradley L, Yaworsky PJ, Walsh FS. Myostatin as a therapeutic target for musculoskeletal disease. Cell Mol Life Sci. 2008 Jul;65(14):2119-24. PMID:18425412 doi:10.1007/s00018-008-8077-3
- ↑ McPherron AC, Lawler AM, Lee SJ. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature. 1997 May 1;387(6628):83-90. PMID:9139826 doi:10.1038/387083a0
- ↑ Wolfman NM, McPherron AC, Pappano WN, Davies MV, Song K, Tomkinson KN, Wright JF, Zhao L, Sebald SM, Greenspan DS, Lee SJ. Activation of latent myostatin by the BMP-1/tolloid family of metalloproteinases. Proc Natl Acad Sci U S A. 2003 Dec 23;100(26):15842-6. Epub 2003 Dec 11. PMID:14671324 doi:10.1073/pnas.2534946100
- ↑ Nakamura T, Takio K, Eto Y, Shibai H, Titani K, Sugino H. Activin-binding protein from rat ovary is follistatin. Science. 1990 Feb 16;247(4944):836-8. PMID:2106159