Caspase-3 Regulatory Mechanisms
From Proteopedia
(→Caspase-3 Active Site and Loop Bundle Analysis) |
(→Caspase-3 Active Site and Loop Bundle Analysis) |
||
| Line 29: | Line 29: | ||
| - | <scene name='Caspase-3_Regulatory_Mechanisms/ | + | <scene name='Caspase-3_Regulatory_Mechanisms/Scene2_nospin_labels/1'>L2 and L2'</scene> |
| - | + | ||
GAAAAHHHHHHHH | GAAAAHHHHHHHH | ||
| + | |||
Revision as of 17:29, 12 December 2012
Introduction
Caspases are cysteine-dependent aspartic acid proteases and are the key facilitators of apoptosis or programmed cell death. Apoptosis is tightly regulated by these caspases, and dysregulation of caspase functions have been implicated in wide variety of diseases such as neurodegeneration, cancer, heart disease and some metabolic disorders. As such, caspases are considered to be attractive drug targets to treat these disorders.
Existing as proenzymes, caspases undergo proteolytic processing at conserved aspartate residues in their intersubunit linker to produce the large and small subunit. These subunits then dimerize to form the active enzyme. Any apoptotic signal received by the cell results in sequential activation of caspases. Upstream or initator caspases (-2,-8, -9 and -10) are first activated by forming a holoenzyme wherein they associate with another protein platform or adaptor protein. Once active, initiator caspases cleave and activate the executioner caspases (-3, -6 and -7) which in turn cleave their respective protein targets initiating cell death.
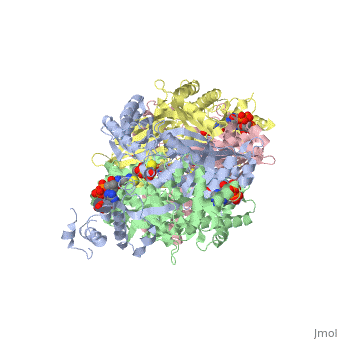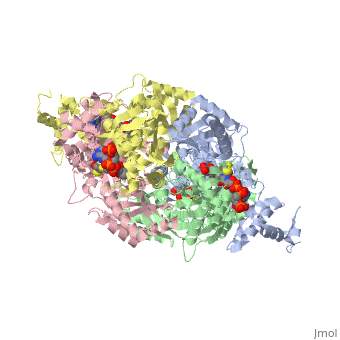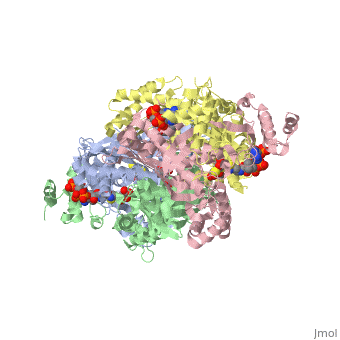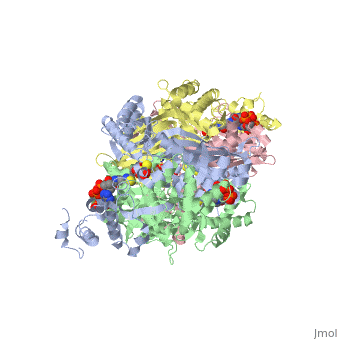
Caspase-3 structure
Regulation of Caspase-3
| |||||||||||
Caspase-3 Active Site and Loop Bundle Analysis
Lets give it a shot.
| |||||||||||
Proteopedia Page Contributors and Editors (what is this?)
Scott Eron, Banyuhay P. Serrano, Alexander Berchansky, Yunlong Zhao, Jaime Prilusky, Michal Harel