This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 714
From Proteopedia
| Line 7: | Line 7: | ||
== Overview == | == Overview == | ||
| - | |||
| - | == Structure == | ||
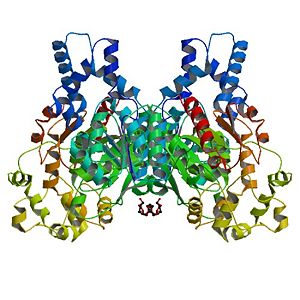
<Structure load='1s8o_mm1.pdb' size='400' frame='true' align='left' caption='X-ray crystal structure of hsEH' scene='Insert optional scene name here' /> | <Structure load='1s8o_mm1.pdb' size='400' frame='true' align='left' caption='X-ray crystal structure of hsEH' scene='Insert optional scene name here' /> | ||
| + | |||
| + | == Structure == | ||
The Human soluble Epoxide hydrolase is a homodimer. Each subunit has <scene name='Sandbox_Reserved_714/Catalytic_domains/1'>two catalytic domains</scene>, linked by a proline-rich section. | The Human soluble Epoxide hydrolase is a homodimer. Each subunit has <scene name='Sandbox_Reserved_714/Catalytic_domains/1'>two catalytic domains</scene>, linked by a proline-rich section. | ||
| + | The C-terminal domain is called Cytosolic epoxide hydrolase 2: it catalyzes the trans-addition of water to epoxides in order to product glycols. The <scene name='Sandbox_Reserved_714/Cter_activesite/1'>active site</scene> is made of five residues. The two tyrosines (Y383 and Y466) plays a role. | ||
Revision as of 19:10, 30 December 2012
Contents |
Overview
|
Structure
The Human soluble Epoxide hydrolase is a homodimer. Each subunit has , linked by a proline-rich section. The C-terminal domain is called Cytosolic epoxide hydrolase 2: it catalyzes the trans-addition of water to epoxides in order to product glycols. The is made of five residues. The two tyrosines (Y383 and Y466) plays a role.
Mechanism
Inhibitors
External ressources
References
Proteopedia Page Contributors and Editors
DUTREUX Fabien, BONHOURE Anna

