This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 714
From Proteopedia
(Difference between revisions)
| Line 17: | Line 17: | ||
== Mechanism == | == Mechanism == | ||
| + | |||
| + | === C-terminal domain === | ||
The C-terminal domain is called Cytosolic epoxide hydrolase 2: it catalyzes the trans-addition of water to epoxides in order to product glycols<ref>PMID:15822179</ref>. The <scene name='Sandbox_Reserved_714/Cter_activesite/3'>active site</scene> is made of five residues. The 3D structure of this active site is maintained by hydrogen bonds, including those created by D496. The two tyrosines (Y383 and Y466) assist the proper positioning of the substrate by polarizing it, thanks to their hydroxyl groups. D335 acts as a nucleophilic acid. Finally, H524 plays the role of a base in order to release the final product. | The C-terminal domain is called Cytosolic epoxide hydrolase 2: it catalyzes the trans-addition of water to epoxides in order to product glycols<ref>PMID:15822179</ref>. The <scene name='Sandbox_Reserved_714/Cter_activesite/3'>active site</scene> is made of five residues. The 3D structure of this active site is maintained by hydrogen bonds, including those created by D496. The two tyrosines (Y383 and Y466) assist the proper positioning of the substrate by polarizing it, thanks to their hydroxyl groups. D335 acts as a nucleophilic acid. Finally, H524 plays the role of a base in order to release the final product. | ||
| Line 22: | Line 24: | ||
The reaction proceeds in two steps, including the formation of a covalent intermediate. | The reaction proceeds in two steps, including the formation of a covalent intermediate. | ||
First, the substrate (epoxide) is accepted in the active site and its oxygen forms hydrogen bonds with Y383 and Y466. The oxygen of D335 attacks one of the two carbons included in the epoxide function. As a result, the oxygen of the subtrate takes the hydrogen of the hydroxyl function of Y466: the covalent intermediate is formed, and linked to D335. | First, the substrate (epoxide) is accepted in the active site and its oxygen forms hydrogen bonds with Y383 and Y466. The oxygen of D335 attacks one of the two carbons included in the epoxide function. As a result, the oxygen of the subtrate takes the hydrogen of the hydroxyl function of Y466: the covalent intermediate is formed, and linked to D335. | ||
| + | |||
| + | === N-terminal domain === | ||
The N-terminal domain is responsible of the Mg<sup>2+</sup> dependant hydrolysis of p-nitrophenyl phosphate <ref>PMID:15096040</ref>. Its <scene name='Sandbox_Reserved_714/Nter_activesite/1'>active site</scene> contains several conserved aspartates in phosphatases and phosphonatases: D9, D11, D184 and D185. This enzymatic activity is Mg<sup>2+</sup> dependant, because the structure of the active site is in its optimal conformation when the cation makes coordination interactions. When the catalytic activity of the N-term domain is available, Magnesium is octahedrally coordinated with the four aspartates, one water molecule and the phosphate belonging to the substrate. | The N-terminal domain is responsible of the Mg<sup>2+</sup> dependant hydrolysis of p-nitrophenyl phosphate <ref>PMID:15096040</ref>. Its <scene name='Sandbox_Reserved_714/Nter_activesite/1'>active site</scene> contains several conserved aspartates in phosphatases and phosphonatases: D9, D11, D184 and D185. This enzymatic activity is Mg<sup>2+</sup> dependant, because the structure of the active site is in its optimal conformation when the cation makes coordination interactions. When the catalytic activity of the N-term domain is available, Magnesium is octahedrally coordinated with the four aspartates, one water molecule and the phosphate belonging to the substrate. | ||
Revision as of 15:17, 2 January 2013
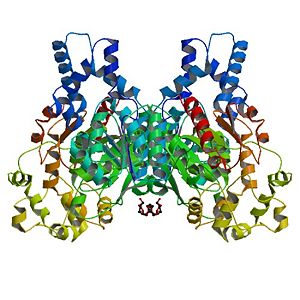
Human Soluble Epoxide Hydrolase: Biological assembly, 1s8o
Contents |
Overview
| |||||||||||
External ressources
References
- ↑ Morisseau C, Hammock BD. Epoxide hydrolases: mechanisms, inhibitor designs, and biological roles. Annu Rev Pharmacol Toxicol. 2005;45:311-33. PMID:15822179 doi:10.1146/annurev.pharmtox.45.120403.095920
- ↑ Gomez GA, Morisseau C, Hammock BD, Christianson DW. Structure of human epoxide hydrolase reveals mechanistic inferences on bifunctional catalysis in epoxide and phosphate ester hydrolysis. Biochemistry. 2004 Apr 27;43(16):4716-23. PMID:15096040 doi:10.1021/bi036189j
Proteopedia Page Contributors and Editors
DUTREUX Fabien, BONHOURE Anna

