This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
2hkq
From Proteopedia
| Line 4: | Line 4: | ||
==Overview== | ==Overview== | ||
| - | Dynamic microtubule plus-end tracking protein (+TIP) networks are | + | Dynamic microtubule plus-end tracking protein (+TIP) networks are implicated in all functions of microtubules, but the molecular determinants of their interactions are largely unknown. Here, we have explored key binding modes of +TIPs by analyzing the interactions between selected CAP-Gly, EB-like, and carboxy-terminal EEY/F-COO(-) sequence motifs. X-ray crystallography and biophysical binding studies demonstrate that the beta2-beta3 loop of CAP-Gly domains determines EB-like motif binding specificity. They further show how CAP-Gly domains serve as recognition domains for EEY/F-COO(-) motifs, which represent characteristic and functionally important sequence elements in EB, CLIP-170, and alpha-tubulin. Our findings provide a molecular basis for understanding the modular interaction modes between alpha-tubulin, CLIPs, EB proteins, and the dynactin-dynein motor complex and suggest that multiple low-affinity binding sites in different combinations control dynamic +TIP networks at microtubule ends. They further offer insights into the structural consequences of genetic CAP-Gly domain defects found in severe human disorders. |
==Disease== | ==Disease== | ||
| Line 17: | Line 17: | ||
[[Category: Protein complex]] | [[Category: Protein complex]] | ||
[[Category: Honnappa, S.]] | [[Category: Honnappa, S.]] | ||
| - | [[Category: Steinmetz, M | + | [[Category: Steinmetz, M O.]] |
| - | [[Category: Winkler, F | + | [[Category: Winkler, F K.]] |
[[Category: +tip protein complex structure]] | [[Category: +tip protein complex structure]] | ||
[[Category: coiled coil]] | [[Category: coiled coil]] | ||
| Line 27: | Line 27: | ||
[[Category: p150glued]] | [[Category: p150glued]] | ||
| - | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Feb 21 17:42:54 2008'' |
Revision as of 15:42, 21 February 2008
|
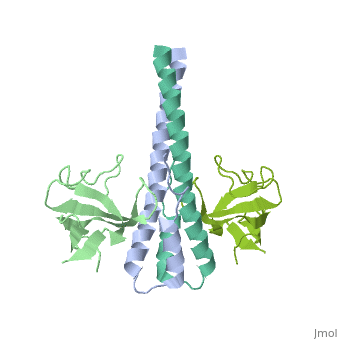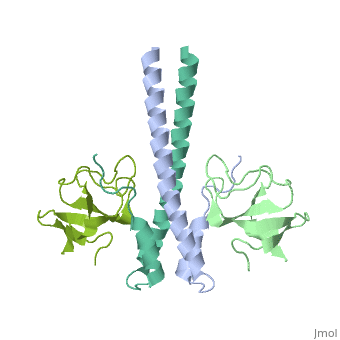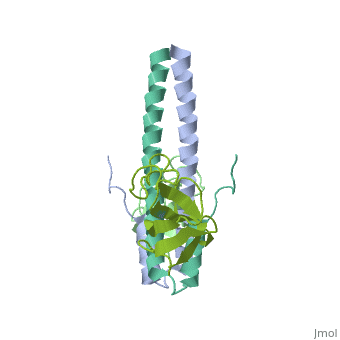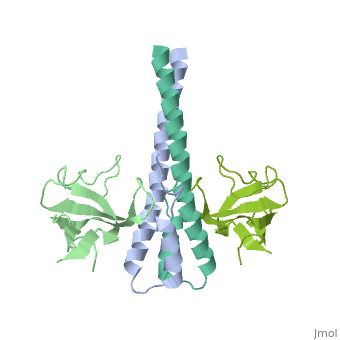
Crystal structure of the C-terminal domain of human EB1 in complex with the CAP-Gly domain of human Dynactin-1 (p150-Glued)
Contents |
Overview
Dynamic microtubule plus-end tracking protein (+TIP) networks are implicated in all functions of microtubules, but the molecular determinants of their interactions are largely unknown. Here, we have explored key binding modes of +TIPs by analyzing the interactions between selected CAP-Gly, EB-like, and carboxy-terminal EEY/F-COO(-) sequence motifs. X-ray crystallography and biophysical binding studies demonstrate that the beta2-beta3 loop of CAP-Gly domains determines EB-like motif binding specificity. They further show how CAP-Gly domains serve as recognition domains for EEY/F-COO(-) motifs, which represent characteristic and functionally important sequence elements in EB, CLIP-170, and alpha-tubulin. Our findings provide a molecular basis for understanding the modular interaction modes between alpha-tubulin, CLIPs, EB proteins, and the dynactin-dynein motor complex and suggest that multiple low-affinity binding sites in different combinations control dynamic +TIP networks at microtubule ends. They further offer insights into the structural consequences of genetic CAP-Gly domain defects found in severe human disorders.
Disease
Known diseases associated with this structure: Amyotrophic lateral sclerosis, susceptibility to OMIM:[601143], Neuropathy, distal hereditary motor, type VIIB OMIM:[601143]
About this Structure
2HKQ is a Protein complex structure of sequences from Homo sapiens. Full crystallographic information is available from OCA.
Reference
Key interaction modes of dynamic +TIP networks., Honnappa S, Okhrimenko O, Jaussi R, Jawhari H, Jelesarov I, Winkler FK, Steinmetz MO, Mol Cell. 2006 Sep 1;23(5):663-71. PMID:16949363
Page seeded by OCA on Thu Feb 21 17:42:54 2008