This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox FEBS Gdansk 06
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
== Structure of HtpG == | == Structure of HtpG == | ||
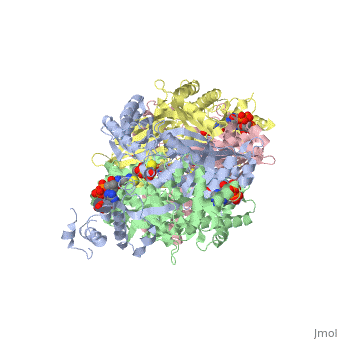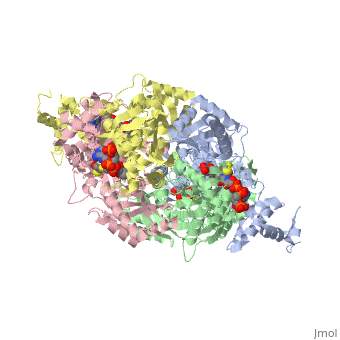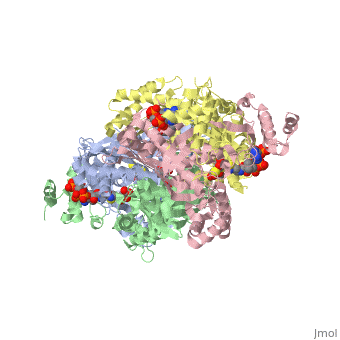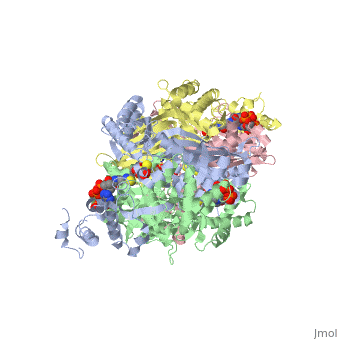
<StructureSection load='1dq8' size='350' side='right' caption='Structure of HMG-CoA reductase (PDB entry [[1dq8]])' scene=''> | <StructureSection load='1dq8' size='350' side='right' caption='Structure of HMG-CoA reductase (PDB entry [[1dq8]])' scene=''> | ||
| - | + | In eukaryotes, the ubiquitous and abundant members of the 90 kilodalton heat-shock protein (hsp90) chaperone family facilitate the folding and conformational changes of a broad array of proteins important in cell signaling, proliferation, and survival. Here we describe the effects of nucleotides on the structure of full-length HtpG, the Escherichia coli hsp90 ortholog | |
</StructureSection> | </StructureSection> | ||
Revision as of 11:15, 13 July 2013
Structure of HtpG
| |||||||||||
|