This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
InhA
From Proteopedia
(Difference between revisions)
| Line 39: | Line 39: | ||
=InhA and Thioamide Drugs= | =InhA and Thioamide Drugs= | ||
| - | + | [[Image:ETH, EAD, PTH, P1H structures.png|thumb|left|upright=1.5|alt=ETH, EAD, PTH, and P1H.|Fig 3: Structures of ETH, EAD, PTH, and P1H]] | |
| - | + | ||
| - | [[Image:ETH, EAD, PTH, P1H structures.png|thumb| | + | |
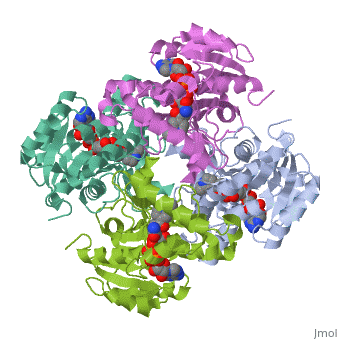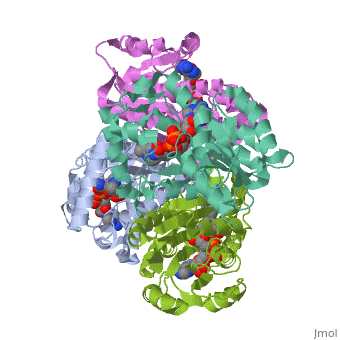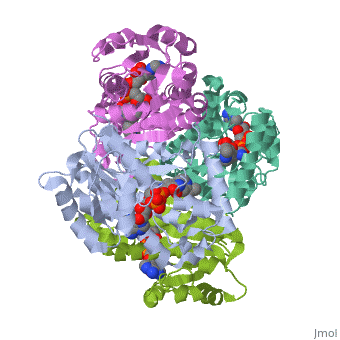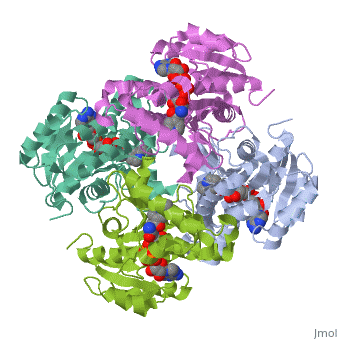
The primary target of the thioamide drugs PTH and ETH have been shown to be InhA <scene name='Sandbox_Reserved_321/Structural_progresion/1'>(go to original scene)</scene> in both genetic and molecular experiments<ref name ="mech of thioamide drug action"/>. Both PTH and ETH require activation by various cellular componets to form the NAD adduct that acts to inhibit InhA, and therefore connot be studied in [http://en.wikipedia.org/wiki/In_vitro in vitro]<ref name ="mech of thioamide drug action"/>. The exacct mechanism of their activation is still under speculation, however a flavin monooxygenase (EthA) has been shown to participate in ETH and PTH activation<ref name ="mech of thioamide drug action"/>. In fact, strains of ''M. tuberculosis'' that have mutations in the gene which express EthA exhibit resistance to thioamide drugs<ref name ="mech of thioamide drug action"/>. Currently studies are being carried out to determine other methods of treatment for mycobaterial infections that dont require activation by cellular constituents, due to the incerease of drug resistant cases world wide. | The primary target of the thioamide drugs PTH and ETH have been shown to be InhA <scene name='Sandbox_Reserved_321/Structural_progresion/1'>(go to original scene)</scene> in both genetic and molecular experiments<ref name ="mech of thioamide drug action"/>. Both PTH and ETH require activation by various cellular componets to form the NAD adduct that acts to inhibit InhA, and therefore connot be studied in [http://en.wikipedia.org/wiki/In_vitro in vitro]<ref name ="mech of thioamide drug action"/>. The exacct mechanism of their activation is still under speculation, however a flavin monooxygenase (EthA) has been shown to participate in ETH and PTH activation<ref name ="mech of thioamide drug action"/>. In fact, strains of ''M. tuberculosis'' that have mutations in the gene which express EthA exhibit resistance to thioamide drugs<ref name ="mech of thioamide drug action"/>. Currently studies are being carried out to determine other methods of treatment for mycobaterial infections that dont require activation by cellular constituents, due to the incerease of drug resistant cases world wide. | ||
| Line 76: | Line 74: | ||
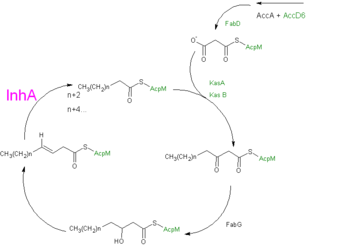
InhA can be further classified into the acyl carrier protein family(ACP's). These proteins generally all function in the transport of substrates in a myriad of pathways, such as: the synthesis of polypeptides and fatty acids<ref name ="Acyl Carrier Proteins">PMID:17012233</ref>. | InhA can be further classified into the acyl carrier protein family(ACP's). These proteins generally all function in the transport of substrates in a myriad of pathways, such as: the synthesis of polypeptides and fatty acids<ref name ="Acyl Carrier Proteins">PMID:17012233</ref>. | ||
| + | </StructureSection> | ||
| + | __NOTOC__ | ||
''3D structures of ACP family'' | ''3D structures of ACP family'' | ||
Revision as of 07:48, 21 August 2013
| |||||||||||
3D structures of ACP family
Enoyl-Acyl-Carrier Protein Reductase
Additional Resources
- Mycobacterium tuberculosis InhA bound with ETH-NAD adduct, in the RCSB Protein Data Bank
- Mycobacterium tuberculosis InhA bound with PTH-NAD adduct, in the RCSB Protein Data Bank
- Crystal structure of wild-type InhA:NADH complex, in the RCSB Protein Data Bank
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Sacchettini, James (New Rochelle, NY) 1999 INHA crystals and three dimensional structure United States Albert Einstein College of Medicine of Yeshiva University (Bronx, NY) 5882878 http://www.freepatentsonline.com/5882878.html
- ↑ 2.0 2.1 2.2 Molle V, Gulten G, Vilcheze C, Veyron-Churlet R, Zanella-Cleon I, Sacchettini JC, Jacobs WR Jr, Kremer L. Phosphorylation of InhA inhibits mycolic acid biosynthesis and growth of Mycobacterium tuberculosis. Mol Microbiol. 2010 Dec;78(6):1591-605. doi:, 10.1111/j.1365-2958.2010.07446.x. Epub 2010 Nov 9. PMID:21143326 doi:10.1111/j.1365-2958.2010.07446.x
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 Wang F, Langley R, Gulten G, Dover LG, Besra GS, Jacobs WR Jr, Sacchettini JC. Mechanism of thioamide drug action against tuberculosis and leprosy. J Exp Med. 2007 Jan 22;204(1):73-8. Epub 2007 Jan 16. PMID:17227913 doi:10.1084/jem.20062100
- ↑ . PMID:216315890657
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 5.7 Dias MV, Vasconcelos IB, Prado AM, Fadel V, Basso LA, de Azevedo WF Jr, Santos DS. Crystallographic studies on the binding of isonicotinyl-NAD adduct to wild-type and isoniazid resistant 2-trans-enoyl-ACP (CoA) reductase from Mycobacterium tuberculosis. J Struct Biol. 2007 Sep;159(3):369-80. Epub 2007 May 3. PMID:17588773 doi:http://dx.doi.org/10.1016/j.jsb.2007.04.009
- ↑ 6.0 6.1 Rozwarski DA, Vilcheze C, Sugantino M, Bittman R, Sacchettini JC. Crystal structure of the Mycobacterium tuberculosis enoyl-ACP reductase, InhA, in complex with NAD+ and a C16 fatty acyl substrate. J Biol Chem. 1999 May 28;274(22):15582-9. PMID:10336454
- ↑ Wilson M, DeRisi J, Kristensen HH, Imboden P, Rane S, Brown PO, Schoolnik GK. Exploring drug-induced alterations in gene expression in Mycobacterium tuberculosis by microarray hybridization. Proc Natl Acad Sci U S A. 1999 Oct 26;96(22):12833-8. PMID:10536008
- ↑ 8.0 8.1 Gurvitz A, Hiltunen JK, Kastaniotis AJ. Function of heterologous Mycobacterium tuberculosis InhA, a type 2 fatty acid synthase enzyme involved in extending C20 fatty acids to C60-to-C90 mycolic acids, during de novo lipoic acid synthesis in Saccharomyces cerevisiae. Appl Environ Microbiol. 2008 Aug;74(16):5078-85. Epub 2008 Jun 13. PMID:18552191 doi:10.1128/AEM.00655-08
- ↑ 9.0 9.1 Bhatt A, Brown AK, Singh A, Minnikin DE, Besra GS. Loss of a mycobacterial gene encoding a reductase leads to an altered cell wall containing beta-oxo-mycolic acid analogs and accumulation of ketones. Chem Biol. 2008 Sep 22;15(9):930-9. PMID:18804030 doi:10.1016/j.chembiol.2008.07.007
- ↑ 10.0 10.1 10.2 Parikh S, Moynihan DP, Xiao G, Tonge PJ. Roles of tyrosine 158 and lysine 165 in the catalytic mechanism of InhA, the enoyl-ACP reductase from Mycobacterium tuberculosis. Biochemistry. 1999 Oct 12;38(41):13623-34. PMID:10521269
- ↑ Oppermann U, Filling C, Hult M, Shafqat N, Wu X, Lindh M, Shafqat J, Nordling E, Kallberg Y, Persson B, Jornvall H. Short-chain dehydrogenases/reductases (SDR): the 2002 update. Chem Biol Interact. 2003 Feb 1;143-144:247-53. PMID:12604210
- ↑ Rafi S, Novichenok P, Kolappan S, Zhang X, Stratton CF, Rawat R, Kisker C, Simmerling C, Tonge PJ. Structure of acyl carrier protein bound to FabI, the FASII enoyl reductase from Escherichia coli. J Biol Chem. 2006 Dec 22;281(51):39285-93. Epub 2006 Sep 29. PMID:17012233 doi:10.1074/jbc.M608758200