This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 795
From Proteopedia
(Difference between revisions)
| Line 4: | Line 4: | ||
==Introduction and General Structure== | ==Introduction and General Structure== | ||
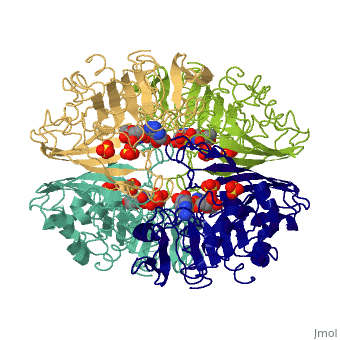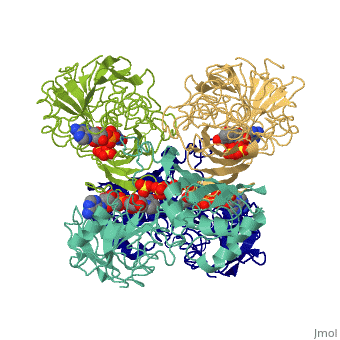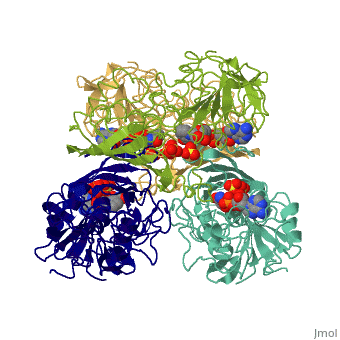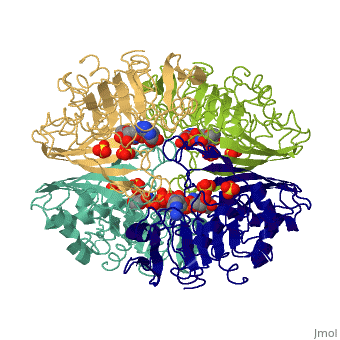
| - | + | <Structure load='3gpd' size='500' frame='true' align='right' caption='Glyceraldehyde-3-phosphate dehydrogenase' scene='Insert optional scene name here' /> Glyceraldehyde 3-phosphate dehydrogenase, or G-3-P dehydrogenase, is an enzyme that plays a significant role in metabolism. It catalyzes a step in glycolysis in which glyceraldehyde 3-phosphate is converted to 3-phospho-D-glyceroyl phosphate. G-3-P dehydrogenase is a tetramer protein made up of four subunits. The <scene name='56/563207/Secondary_structure/1'>secondary structure</scene> of G-3-P dehydrogenase consists of alpha helices which are displayed in purple and beta sheets which are displayed in green. The beta sheets are parallel. | |
==Hydrogen Bonds== | ==Hydrogen Bonds== | ||
The <scene name='56/563207/Hydrogen_bonding/2'>hydrogen bonding</scene> of the backbone is displayed in green. There are no disulfide bonds present in this enzyme. | The <scene name='56/563207/Hydrogen_bonding/2'>hydrogen bonding</scene> of the backbone is displayed in green. There are no disulfide bonds present in this enzyme. | ||
Revision as of 02:39, 15 October 2013
| This Sandbox is Reserved from Oct 10, 2013, through May 20, 2014 for use in the course "CHEM 410 Biochemistry 1 and 2" taught by Hanna Tims at the Messiah College. This reservation includes Sandbox Reserved 780 through Sandbox Reserved 807. |
To get started:
More help: Help:Editing |
Introduction and General Structure
|
Hydrogen Bonds
The of the backbone is displayed in green. There are no disulfide bonds present in this enzyme.