User:Andrew Wills/Sandbox 1
From Proteopedia
| Line 16: | Line 16: | ||
==Active Sites and Mechanism== | ==Active Sites and Mechanism== | ||
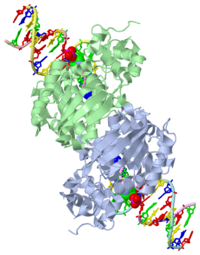
| - | The HhH segment of AlkA connects to the phosphodiester backbone of DNA by hydrogen bonding and sodium | + | The HhH segment of AlkA connects to the phosphodiester backbone of DNA by hydrogen bonding and <scene name='56/566536/Na_ion/1'>sodium-metal ion</scene> interactions. The binding of the HhH segment to DNA stabilizes the damaged base and creates a 66 degree bend away from the protein that widens the minor groove of DNA. Leu 125 fits into the minor groove between base pairs and allows the alkylated base to be flipped into the active site (Hollis). |
The AlkA active site is located where the second and third domains are separated by a deep nonpolar cleft that is lined with the aromatic side chains Phe18, Tyr273, Trp272, Tyr222, and Trp218 (Labahn). These side chains create a DNA binding pocket for the alkylated base (Labahn). Once the damaged base is in the active site, Trp272 stabilizes the flipped out alkylated base in the binding pocket by aromatic ring-stacking interactions (Moe). Asp238 is essential for allowing the reaction to proceed, and points into the nonpolar pocket in order to allow stabilization of a carbocation intermediate. This stabilization is what allows the cleavage of the glycosidic bond on the damaged base (Moe). Mutation or inhibition of the Asp238 residue will prevent AlkA from performing base excision repair. | The AlkA active site is located where the second and third domains are separated by a deep nonpolar cleft that is lined with the aromatic side chains Phe18, Tyr273, Trp272, Tyr222, and Trp218 (Labahn). These side chains create a DNA binding pocket for the alkylated base (Labahn). Once the damaged base is in the active site, Trp272 stabilizes the flipped out alkylated base in the binding pocket by aromatic ring-stacking interactions (Moe). Asp238 is essential for allowing the reaction to proceed, and points into the nonpolar pocket in order to allow stabilization of a carbocation intermediate. This stabilization is what allows the cleavage of the glycosidic bond on the damaged base (Moe). Mutation or inhibition of the Asp238 residue will prevent AlkA from performing base excision repair. | ||
Revision as of 02:45, 4 November 2013
| |||||||||
| 1diz, resolution 2.50Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligands: | |||||||||
| Non-Standard Residues: | |||||||||
| Activity: | DNA-3-methyladenine glycosylase II, with EC number 3.2.2.21 | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, PDBsum, RCSB | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Contents |
CRYSTAL STRUCTURE OF E. COLI 3-METHYLADENINE DNA GLYCOSYLASE (ALKA) COMPLEXED WITH DNA
General Function
1diz Escherichia coli 3 methyladenine DNA glycosylase II (AlkA) is a DNA repair enzyme that initiates base excision repair for the removal of alkylated bases. Aflatoxin B1 is an example of a toxin that attacks guanine and adenine at their N-7 atom to form alkylated bases (stryker), which prevent regulatory proteins from binding to DNA and blocks replicative polymerases (Hollis). AlkA initiates base excision repair by first locating and binding to the alkylated DNA. It then flips the affected base out of the DNA double helix and into the active site of the enzyme. Once in the active site, AlkA hydrolyzes the glycosidic bond to release the damaged base and leave the sugar phosphate backbone intact. This creates the AP site that is either devoid of a purine or pyridine. The AP site signals to other base excision repair enzymes to insert an undamaged nucleotide based on the undamaged complementary strand and seal the DNA.
| |||||||||||
See Also
Reference
- Hollis T, Ichikawa Y, Ellenberger T. DNA bending and a flip-out mechanism for base excision by the helix-hairpin-helix DNA glycosylase, Escherichia coli AlkA. EMBO J. 2000 Feb 15;19(4):758-66. PMID:10675345 doi:http://dx.doi.org/10.1093/emboj/19.4.758