This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Ku protein
From Proteopedia
(Difference between revisions)
| Line 23: | Line 23: | ||
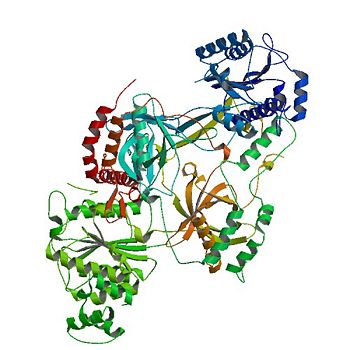
Unlike other DNA binding proteins, the Ku protein is asymmetrical from the differences between the Ku70 and Ku80 subunits. | Unlike other DNA binding proteins, the Ku protein is asymmetrical from the differences between the Ku70 and Ku80 subunits. | ||
This asymmetry leads to different favorable locations for DNA based on major and minor grooves.<ref name="Walker"/> | This asymmetry leads to different favorable locations for DNA based on major and minor grooves.<ref name="Walker"/> | ||
| - | The Ku70 subunit is <scene name='56/567269/Ku70_subunit/ | + | The Ku70 subunit is <scene name='56/567269/Ku70_subunit/5'>angled closer</scene> to DNA at the double strand break, providing protection and interaction with its domains.<ref name="source2"> PMID: 19715578</ref> |
In contrast, the Ku80 subunit <scene name='56/567269/Ku80_subunit/4'>associates with</scene> DNA away from the free end.<ref name="Walker"/> Once a homodimer, the protein has diverged into two domains that are now 15% similar in residues. <ref name="source3"> PMID: 9663392</ref> | In contrast, the Ku80 subunit <scene name='56/567269/Ku80_subunit/4'>associates with</scene> DNA away from the free end.<ref name="Walker"/> Once a homodimer, the protein has diverged into two domains that are now 15% similar in residues. <ref name="source3"> PMID: 9663392</ref> | ||
Revision as of 14:33, 5 November 2013
| |||||||||||
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 Walker JR, Corpina RA, Goldberg J. Structure of the Ku heterodimer bound to DNA and its implications for double-strand break repair. Nature. 2001 Aug 9;412(6847):607-14. PMID:11493912 doi:10.1038/35088000
- ↑ Bennett SM, Neher TM, Shatilla A, Turchi JJ. Molecular analysis of Ku redox regulation. BMC Mol Biol. 2009 Aug 28;10:86. doi: 10.1186/1471-2199-10-86. PMID:19715578 doi:http://dx.doi.org/10.1186/1471-2199-10-86
- ↑ 3.0 3.1 3.2 Polotnianka RM, Li J, Lustig AJ. The yeast Ku heterodimer is essential for protection of the telomere against nucleolytic and recombinational activities. Curr Biol. 1998 Jul 2;8(14):831-4. PMID:9663392
- ↑ 4.0 4.1 Bertuch AA, Lundblad V. The Ku heterodimer performs separable activities at double-strand breaks and chromosome termini. Mol Cell Biol. 2003 Nov;23(22):8202-15. PMID:14585978