Glutamate receptor (GluA2)
From Proteopedia
(→See Also) |
(RESTORED THE EXTRA WINDOWS TO SHOW SYMMETRY - THIS CANNOT BE ONE STRUCTURE SECTION BECAUSE IT IS SO COMPLEX) |
||
| Line 1: | Line 1: | ||
__NOTOC__ | __NOTOC__ | ||
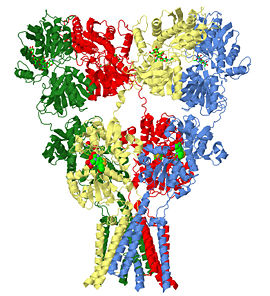
| - | + | The glutamate receptor is the ion channel opened by glutamate that keeps neurons in touch by mediating fast cell-to-cell information transfer in the nervous system. Several studies have revealed structures for portions of the glutamate receptor <ref name="r80">PMID: 19461580</ref><ref name="r14">PMID: 19465914</ref><ref name="r22">PMID: 19910922</ref><ref>PMID: 9804426</ref>. Groundbreaking work elucidated the structure of a complete functional, homomeric glutamate receptor<ref name="main">PMID:19946266</ref><ref>PMID: 20010675</ref> and that structure, [[3kg2]], is the subject of this page. | |
| - | + | ||
| - | The | + | |
[[Image:3KG2-snapshot-900x900-14724.jpg|left|270px]] | [[Image:3KG2-snapshot-900x900-14724.jpg|left|270px]] | ||
| - | {{clear}} | ||
==Structure of the Glutamate Receptor (GluA2)== | ==Structure of the Glutamate Receptor (GluA2)== | ||
| - | + | <StructureSection load='3kg2' size='500' side='right' scene='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Default3kg2/1' caption='Glutamate Receptor ([[3kg2]])'> | |
===Overview=== | ===Overview=== | ||
The homomeric rat GluA2 receptor <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Default3kg2/1'>has four subunits</scene> arranged in a 'Y'-shape with the <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Meas3kg2/1'>'top' being about three times the width of the 'bottom'</scene><ref name="main" />. This structure is a functional homotetramer of the AMPA-subtype; native ionotropic glutamate receptors are almost exclusively heterotetramers. {{Link Toggle FancyCartoonHighQualityView}}. | The homomeric rat GluA2 receptor <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Default3kg2/1'>has four subunits</scene> arranged in a 'Y'-shape with the <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Meas3kg2/1'>'top' being about three times the width of the 'bottom'</scene><ref name="main" />. This structure is a functional homotetramer of the AMPA-subtype; native ionotropic glutamate receptors are almost exclusively heterotetramers. {{Link Toggle FancyCartoonHighQualityView}}. | ||
| Line 12: | Line 9: | ||
===Domains=== | ===Domains=== | ||
The subunits themselves are modular <ref>PMID: 7539962</ref>and the major domains are found in layers in the tetrameric structure. | The subunits themselves are modular <ref>PMID: 7539962</ref>and the major domains are found in layers in the tetrameric structure. | ||
| - | + | <!-- select all; spacefill off; select hetero; color cpk; wireframe 0.35; spacefill 0.4; select zk1; spacefill on; color cpk; --> | |
*The 'top' layer is composed of the <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Atd_domain/4'>amino-terminal domain(ATD)</scene> | *The 'top' layer is composed of the <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Atd_domain/4'>amino-terminal domain(ATD)</scene> | ||
::This <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Atd_gly/2'>extracellular domain is glycosylated</scene>. | ::This <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Atd_gly/2'>extracellular domain is glycosylated</scene>. | ||
| Line 38: | Line 35: | ||
*As explored further in [[#Transmembrane domain architecture and the occluded pore|a later section below]] , the <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Tmd_domain_4fold/2'>symmetry is an overall four-fold for the TMD</scene>. Thus, remarkably, the symmetry switches from an overall two-fold symmetry for the ATD and LBD to four-fold for the TMD. | *As explored further in [[#Transmembrane domain architecture and the occluded pore|a later section below]] , the <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Tmd_domain_4fold/2'>symmetry is an overall four-fold for the TMD</scene>. Thus, remarkably, the symmetry switches from an overall two-fold symmetry for the ATD and LBD to four-fold for the TMD. | ||
| - | + | </StructureSection> | |
===Subunit Non-Equivalence, Transmembrane Domain Architecture and the Occluded Pore=== | ===Subunit Non-Equivalence, Transmembrane Domain Architecture and the Occluded Pore=== | ||
| - | + | <StructureSection load='3kg2' size='500' side='left' scene ='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Default3kg2/1' caption='Glutamate Receptor Structure' name='main2NDwindow'> | |
===Subunit non-equivalence=== | ===Subunit non-equivalence=== | ||
| - | As a result of the swapping and symmetry mismatch, there is subunit non-equivalence; even though all the chains are the same chemically, there are two distinct conformations of the subunits. This means there are two matching pairs of subunits. | + | As a result of the swapping and symmetry mismatch, there is subunit non-equivalence; even though all the chains are the same chemically, there are two distinct conformations of the subunits. This means there are two matching pairs of subunits. The pairs are illustrated below and the morphs referred to below will show in the main window on the left: |
| - | + | <table width='380' align='left' cellpadding='2'><tr><td bgcolor='#eeeeee'><applet load='3kg2' size='190' frame='true' align='left' scene='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Ac3kg2letter/1' caption='A is equivalent to C'/></td><td bgcolor='#eeeeee'><applet load='3kg2' size='190' frame='true' align='left' scene='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Bd3kg2letter/2' caption='B is equivalent to D'/></td></tr></table> | |
| - | + | ||
| - | + | ||
| - | + | ||
* <span style="color:forestgreen">Subunit '''A</span>''' is equivalent to <span style="color:cornflowerblue">Subunit '''C'''</span> (in the small structure window in this section). In the main window, a <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Atocmorph/5' target='main2NDwindow'>morph showing the equivalency of the two subunits by rotating around the axis of their symmetry</scene>. | * <span style="color:forestgreen">Subunit '''A</span>''' is equivalent to <span style="color:cornflowerblue">Subunit '''C'''</span> (in the small structure window in this section). In the main window, a <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Atocmorph/5' target='main2NDwindow'>morph showing the equivalency of the two subunits by rotating around the axis of their symmetry</scene>. | ||
* <span style="color:red">Subunit '''B</span>''' is equivalent to Subunit '''D''' (in the small structure window in this section). <!--<span style="color:#FFFF80">Subunit '''D'''</span>(<--says 'Subunit D' in hard-to-read gold color matching the structure)--> In the main window, a <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Btodmorph/5' target='main2NDwindow'>morph showing the equivalency of the two subunits by rotating around the axis of their symmetry</scene>. | * <span style="color:red">Subunit '''B</span>''' is equivalent to Subunit '''D''' (in the small structure window in this section). <!--<span style="color:#FFFF80">Subunit '''D'''</span>(<--says 'Subunit D' in hard-to-read gold color matching the structure)--> In the main window, a <scene name='User:Wayne_Decatur/Sandbox_Glutamate_receptor/Btodmorph/5' target='main2NDwindow'>morph showing the equivalency of the two subunits by rotating around the axis of their symmetry</scene>. | ||
Revision as of 15:18, 25 November 2013
The glutamate receptor is the ion channel opened by glutamate that keeps neurons in touch by mediating fast cell-to-cell information transfer in the nervous system. Several studies have revealed structures for portions of the glutamate receptor [1][2][3][4]. Groundbreaking work elucidated the structure of a complete functional, homomeric glutamate receptor[5][6] and that structure, 3kg2, is the subject of this page.
Structure of the Glutamate Receptor (GluA2)
| |||||||||||
Subunit Non-Equivalence, Transmembrane Domain Architecture and the Occluded Pore
| |||||||||||
Details of Structure Featured
3kg2 is a 4 chains structure of sequences from Rattus norvegicus. Full crystallographic information is available from OCA. Although it is billed as the first structure of a full-length glutamate receptor, the carboxy-terminal domain is not present in the structure.
Reference for the structure
- Sobolevsky AI, Rosconi MP, Gouaux E. X-ray structure, symmetry and mechanism of an AMPA-subtype glutamate receptor. Nature. 2009 Dec 10;462(7274):745-56. Epub . PMID:19946266 doi:10.1038/nature08624
3D structures of glutamate receptors
Ionotropic Glutamate Receptors
See Also
- 1iiw and 1iit and 1ii5 – Prokaryotic glutamte receptor (Glur0) Apo structure and with various ligands bound, including glutmate [13]. This helped cement the notion the glutamate and potassium receptors share structural similarity and possibly evolutionary ancestry [10][11].
- 1bl8 and 1jq1 and 1jq2 – Streptomyces lividans KcsA potassium channel[12][14]: The M1, M2 and M3 segments of GluA2's ion channel overlap remarkably well with the structurally equivalent portions KcsA.
- Molecular Playground/Glutamate Receptor
- Ligand Binding N-Terminal of Metabotropic Glutamate Receptors
References
- ↑ Jin R, Singh SK, Gu S, Furukawa H, Sobolevsky AI, Zhou J, Jin Y, Gouaux E. Crystal structure and association behaviour of the GluR2 amino-terminal domain. EMBO J. 2009 Jun 17;28(12):1812-23. Epub 2009 May 21. PMID:19461580 doi:10.1038/emboj.2009.140
- ↑ Kumar J, Schuck P, Jin R, Mayer ML. The N-terminal domain of GluR6-subtype glutamate receptor ion channels. Nat Struct Mol Biol. 2009 Jun;16(6):631-8. Epub 2009 May 24. PMID:19465914 doi:10.1038/nsmb.1613
- ↑ Karakas E, Simorowski N, Furukawa H. Structure of the zinc-bound amino-terminal domain of the NMDA receptor NR2B subunit. EMBO J. 2009 Dec 16;28(24):3910-20. Epub . PMID:19910922 doi:10.1038/emboj.2009.338
- ↑ Armstrong N, Sun Y, Chen GQ, Gouaux E. Structure of a glutamate-receptor ligand-binding core in complex with kainate. Nature. 1998 Oct 29;395(6705):913-7. PMID:9804426 doi:10.1038/27692
- ↑ 5.0 5.1 Sobolevsky AI, Rosconi MP, Gouaux E. X-ray structure, symmetry and mechanism of an AMPA-subtype glutamate receptor. Nature. 2009 Dec 10;462(7274):745-56. Epub . PMID:19946266 doi:10.1038/nature08624
- ↑ Wollmuth LP, Traynelis SF. Neuroscience: Excitatory view of a receptor. Nature. 2009 Dec 10;462(7274):729-31. PMID:20010675 doi:10.1038/462729a
- ↑ Wo ZG, Oswald RE. Unraveling the modular design of glutamate-gated ion channels. Trends Neurosci. 1995 Apr;18(4):161-8. PMID:7539962
- ↑ Turski L, Huth A, Sheardown M, McDonald F, Neuhaus R, Schneider HH, Dirnagl U, Wiegand F, Jacobsen P, Ottow E. ZK200775: a phosphonate quinoxalinedione AMPA antagonist for neuroprotection in stroke and trauma. Proc Natl Acad Sci U S A. 1998 Sep 1;95(18):10960-5. PMID:9724812
- ↑ Walters MR, Kaste M, Lees KR, Diener HC, Hommel M, De Keyser J, Steiner H, Versavel M. The AMPA antagonist ZK 200775 in patients with acute ischaemic stroke: a double-blind, multicentre, placebo-controlled safety and tolerability study. Cerebrovasc Dis. 2005;20(5):304-9. Epub 2005 Aug 30. PMID:16131799 doi:10.1159/000087929
- ↑ 10.0 10.1 Wo ZG, Oswald RE. Unraveling the modular design of glutamate-gated ion channels. Trends Neurosci. 1995 Apr;18(4):161-8. PMID:7539962
- ↑ 11.0 11.1 Wood MW, VanDongen HM, VanDongen AM. Structural conservation of ion conduction pathways in K channels and glutamate receptors. Proc Natl Acad Sci U S A. 1995 May 23;92(11):4882-6. PMID:7761417
- ↑ 12.0 12.1 Doyle DA, Morais Cabral J, Pfuetzner RA, Kuo A, Gulbis JM, Cohen SL, Chait BT, MacKinnon R. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science. 1998 Apr 3;280(5360):69-77. PMID:9525859
- ↑ Chen GQ, Cui C, Mayer ML, Gouaux E. Functional characterization of a potassium-selective prokaryotic glutamate receptor. Nature. 1999 Dec 16;402(6763):817-21. PMID:10617203 doi:10.1038/45568
- ↑ Liu YS, Sompornpisut P, Perozo E. Structure of the KcsA channel intracellular gate in the open state. Nat Struct Biol. 2001 Oct;8(10):883-7. PMID:11573095 doi:10.1038/nsb1001-883
Additional Literature and Resources
- For additional information, see: Alzheimer's Disease
- For additional information, see: Membrane Channels & Pumps
- Glutamate Receptor on the cover of Nature
- Glutamate receptor Wikipedia entry
- Glutamate Receptors page at the MRC Centre for Synaptic Plasticity at the University of Bristol
Page started with original page seeded by OCA on Wed Dec 16 11:24:54 2009 for 3kg2.
Proteopedia Page Contributors and Editors (what is this?)
Wayne Decatur, Alexander Berchansky, Michal Harel, David Canner, Nikki Hunter
Categories: Rattus norvegicus | Gouaux, E. | Rosconi, M P. | Sobolevsky, A I. | Alternative splicing | Cell membrane | Glycoprotein | Ion channel | Ion transport | Membrane | Membrane protein | Postsynaptic cell membrane | Receptor | Rna editing | Synapse | Tetramer | Transmembrane | Transport | Neuron | Neurotransmitter | Potassium Channels | RCSB PDB Molecule of the Month | Streptomyces lividans | Cabral, J M. | Chait, B T. | Cohen, S L. | Doyle, D A. | Gulbis, J M. | Kuo, A. | Mackinnon, R. | Pfuetzner, R A. | Integral membrane protein | Potassium channel | Topic Page