This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Pesticidal crystal protein
From Proteopedia
(Difference between revisions)
| Line 5: | Line 5: | ||
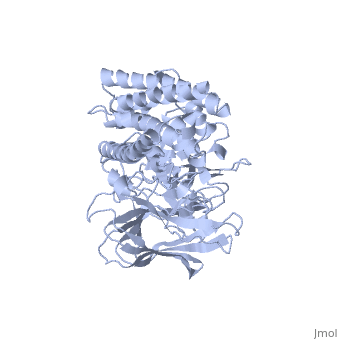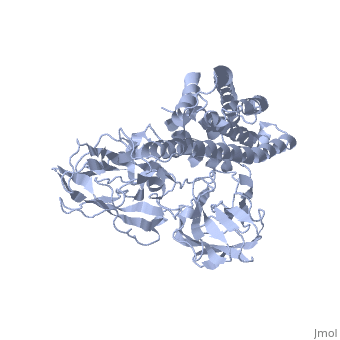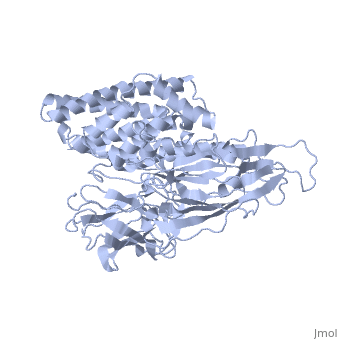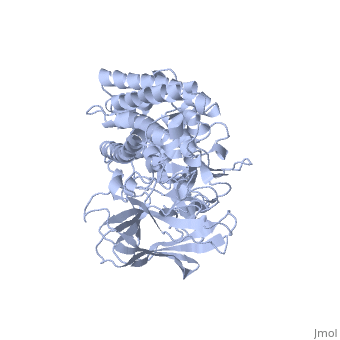
The cry protein has <scene name='57/570587/Three_domains/1'>three domains</scene>: an alpha helical N terminal domain, domain I; a Greek key beta sheet domain III; and a "jelly roll" antiparallel b-sandwich domain III. The core of the protein is the most highly <scene name='57/570587/Conserved_regions/1'>conserved</scene>, with Helix 5 of Domain I and the areas of contact between the domains being the most highly conserved (purple). | The cry protein has <scene name='57/570587/Three_domains/1'>three domains</scene>: an alpha helical N terminal domain, domain I; a Greek key beta sheet domain III; and a "jelly roll" antiparallel b-sandwich domain III. The core of the protein is the most highly <scene name='57/570587/Conserved_regions/1'>conserved</scene>, with Helix 5 of Domain I and the areas of contact between the domains being the most highly conserved (purple). | ||
| - | The insecticidal properties of the Cry protein are associated with domain I, and given the length of the alpha helices and ion conductance data, it is believed that this segment of the protein forms an ion channel. The role of several <scene name='57/570587/Arg_in_domain_i/1'>arginine residues</scene> in Domain I was tested for their role in creating pores in the insect gut membrane via mutation to Ala. | + | The insecticidal properties of the Cry protein are associated with domain I, and given the length of the alpha helices and ion conductance data, it is believed that this segment of the protein forms an ion channel. The role of several <scene name='57/570587/Arg_in_domain_i/1'>arginine residues</scene> in Domain I was tested for their role in creating pores in the insect gut membrane via mutation to Ala. Alternation of Arg 233 lead to almost complete loss of the ion channel formation. This residue interacts with <scene name='57/570587/Arg233_glu_288/1'>Glu 288</scene>, which is in a conserved portion of Domain II. |
</StructureSection> | </StructureSection> | ||
Revision as of 15:29, 2 December 2013
Cry Proteins
| |||||||||||
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Ann Taylor, Alexander Berchansky, Jaime Prilusky