This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 820
From Proteopedia
(Difference between revisions)
| Line 30: | Line 30: | ||
The back-to-back form is stabilized by intermolecular interactions between the | The back-to-back form is stabilized by intermolecular interactions between the | ||
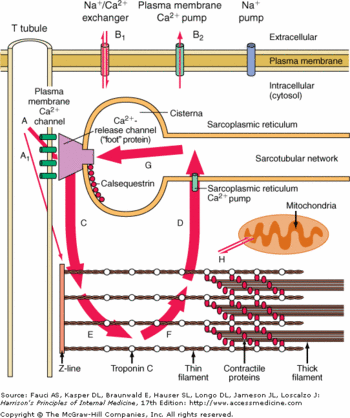
| - | <scene name='56/568018/Oligomere_and_ligand/6'>α4 helix of the domain II</scene> and the | + | <scene name='56/568018/Oligomere_and_ligand/6'>α4 helix of the domain II</scene> and the <scene name='56/568018/Dimer/8'>α3 helix of the domain I</scene> (<scene name='56/568018/Dimer/15'>together</scene>).<ref name="Crystal Structure of calsequestrin from rabbit skeletal muscle sarcoplasmic reticulum (Wang et al., 1998)">http://www.nature.com/nsmb/journal/v5/n6/abs/nsb0698-476.html</ref> The intermolecular salt bridges are built between Glu 215 and Lys 86, Glu 216 and Lys 24, Glu 169 and Lys 85.<ref name="Crystal Structure of calsequestrin from rabbit skeletal muscle sarcoplasmic reticulum (Wang et al., 1998)">http://www.nature.com/nsmb/journal/v5/n6/abs/nsb0698-476.html</ref> There is also a hydrogen bond between Ala 82 and Asn 22. This dimerisation induces a very electronegative pocket at the C-terminal region which enables the binding of Ca2+.<ref name="Crystal Structure of calsequestrin from rabbit skeletal muscle sarcoplasmic reticulum (Wang et al., 1998)">http://www.nature.com/nsmb/journal/v5/n6/abs/nsb0698-476.html</ref> |
| - | <scene name='56/568018/Dimer/8'>α3 helix of the domain I</scene> (<scene name='56/568018/Dimer/15'>together</scene>).<ref name="Crystal Structure of calsequestrin from rabbit skeletal muscle sarcoplasmic reticulum (Wang et al., 1998)">http://www.nature.com/nsmb/journal/v5/n6/abs/nsb0698-476.html</ref> The intermolecular salt bridges are built between Glu 215 and Lys 86, Glu 216 and Lys 24, Glu 169 and Lys 85.<ref name="Crystal Structure of calsequestrin from rabbit skeletal muscle sarcoplasmic reticulum (Wang et al., 1998)">http://www.nature.com/nsmb/journal/v5/n6/abs/nsb0698-476.html</ref> There is also a hydrogen bond between Ala 82 and Asn 22. This dimerisation induces a very electronegative pocket at the C-terminal region which enables the binding of Ca2+.<ref name="Crystal Structure of calsequestrin from rabbit skeletal muscle sarcoplasmic reticulum (Wang et al., 1998)">http://www.nature.com/nsmb/journal/v5/n6/abs/nsb0698-476.html</ref> | + | |
<!--Mettre du VERT --> | <!--Mettre du VERT --> | ||
<!-- Source: Crystal Structure of calsequestrin from rabbit skeletal muscle sarcoplasmic reticulum (Wang et al., 1998) Lien: http://www.nature.com/nsmb/journal/v5/n6/abs/nsb0698-476.html --> | <!-- Source: Crystal Structure of calsequestrin from rabbit skeletal muscle sarcoplasmic reticulum (Wang et al., 1998) Lien: http://www.nature.com/nsmb/journal/v5/n6/abs/nsb0698-476.html --> | ||
Revision as of 10:19, 3 January 2014
| This Sandbox is Reserved from 06/12/2018, through 30/06/2019 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1480 through Sandbox Reserved 1543. |
To get started:
More help: Help:Editing |
| |||||||||||
References
- ↑ Cerrone M, Napolitano C, Priori SG. Catecholaminergic polymorphic ventricular tachycardia: A paradigm to understand mechanisms of arrhythmias associated to impaired Ca(2+) regulation. Heart Rhythm. 2009 Nov;6(11):1652-9. doi: 10.1016/j.hrthm.2009.06.033. Epub 2009 , Jun 30. PMID:19879546 doi:http://dx.doi.org/10.1016/j.hrthm.2009.06.033
- ↑ NCBI Gene Ressource: CASQ2 calsequestrin 2 http://www.ncbi.nlm.nih.gov/gene/845
- ↑ Martin JL. Thioredoxin--a fold for all reasons. Structure. 1995 Mar 15;3(3):245-50. PMID:7788290
- ↑ NCBI Structure Ressource: CASQ2 calsequestrin 2 http://www.ncbi.nlm.nih.gov/Structure/cdd/cddsrv.cgi?ascbin=8&maxaln=10&seltype=2&uid=239372&querygi=429544235&aln=1,227,0,109
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 Crystal Structure of calsequestrin from rabbit skeletal muscle sarcoplasmic reticulum (Wang et al., 1998) http://www.nature.com/nsmb/journal/v5/n6/abs/nsb0698-476.html
- ↑ The asp-rich region at the carboxyl-terminus of calsequestrin binds to Ca2+ and interacts with triadin (Shin et al., 2000) http://www.sciencedirect.com/science/article/pii/S0014579300022468
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 Beard NA, Laver DR, Dulhunty AF. Calsequestrin and the calcium release channel of skeletal and cardiac muscle. Prog Biophys Mol Biol. 2004 May;85(1):33-69. PMID:15050380 doi:http://dx.doi.org/10.1016/j.pbiomolbio.2003.07.001
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 Beard NA, Casarotto MG, Wei L, Varsanyi M, Laver DR, Dulhunty AF. Regulation of ryanodine receptors by calsequestrin: effect of high luminal Ca2+ and phosphorylation. Biophys J. 2005 May;88(5):3444-54. Epub 2005 Feb 24. PMID:15731387 doi:http://dx.doi.org/10.1529/biophysj.104.051441
Proteopedia page contributors and editors
Marc-Antoine JACQUES and Thomas VUILLEMIN