This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox reserved 915
From Proteopedia
(Difference between revisions)
| Line 29: | Line 29: | ||
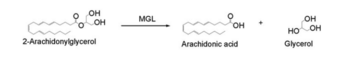
2-AG binds to the catalytic triad and is hydrolyzed. The structure of 2-AG contains a long and flexible aliphatic chain and a polar head that is cleaved. 2-AG is broken down into arachidonic acid and glycerol which makes 2-AG inactive. '''See Overall Reaction.''' | 2-AG binds to the catalytic triad and is hydrolyzed. The structure of 2-AG contains a long and flexible aliphatic chain and a polar head that is cleaved. 2-AG is broken down into arachidonic acid and glycerol which makes 2-AG inactive. '''See Overall Reaction.''' | ||
===Inhibition of Catalytic Triad=== | ===Inhibition of Catalytic Triad=== | ||
| - | Research on MGL is being geared towards inhibiting 2-AG from binding to the catalytic triad and being hydrolyzed. The binding of 2-AG to the catalytic triad can not be inhibited, but it can be extracted before being hydrolyzed. MPD (2-methyl-pentane-2,4-diol)is located at the end of the tunnel where the catalytic triad is at and the tunnel is filled with MPD molecules. MPD being in the same vicinity will extract 2-AG from the triad and the MPD molecule will sit in there in place of 2-AG. This natural inhibition phenomenon is known as interfacial activation. | + | Research on MGL is being geared towards inhibiting 2-AG from binding to the catalytic triad and being hydrolyzed. The binding of 2-AG to the catalytic triad can not be inhibited, but it can be extracted before being hydrolyzed. MPD (2-methyl-pentane-2,4-diol)is located at the end of the tunnel where the catalytic triad is at and the tunnel is filled with MPD molecules. MPD being in the same vicinity will extract 2-AG from the triad and the MPD molecule will sit in there in place of 2-AG. This natural inhibition phenomenon is known as interfacial activation. Inhibition of MGL leads to increase in 2-AG levels since AG is broken down by MGL (Clemente et al. 2012). Through covalent interactions with a Cys residue, NAM, one of the many possible inhibitors, is able to inhibit MGL (Bertrand et al. 2010). |
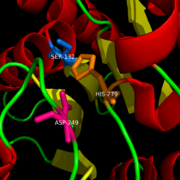
==Ligand Binding Site== | ==Ligand Binding Site== | ||
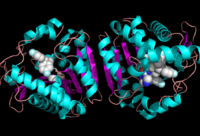
[[Image:Overall_ligand.png|left|200px|thumb|Ligand within the Overall Structure of MGL]] | [[Image:Overall_ligand.png|left|200px|thumb|Ligand within the Overall Structure of MGL]] | ||
| - | The | + | The <scene name='58/580298/Ligand/1'>Ligand binding pocket</scene>of MGL has a large hydrophobic region with a polar bottom (Bertrand et al. 2010). The entrance of the binding pocket for MGL contains a lid, which is very flexible (Bertrand et al. 2010). The binding pocket or tunnel within MGL matches with the overall structure of 2-AG, with 2-AG's polar head being cleaved by the catalytic triad (Bertrand et al. 2010). Bertrand found that in MGL the binding pocket is not adjusted to the ligand's shape. However, the main movements of MGL associated with ligand binding involved the lid region (Bertrand et al. 2010) |
==Overall Reaction== | ==Overall Reaction== | ||
[[Image:Reaction.PNG|350px|thumb|Overall reaction representing the hydrolysis of 2-AG by MGL]] | [[Image:Reaction.PNG|350px|thumb|Overall reaction representing the hydrolysis of 2-AG by MGL]] | ||
Revision as of 20:15, 31 March 2014
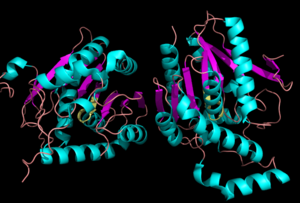
Monoglyceride Lipase (MGL)
| |||||||||||