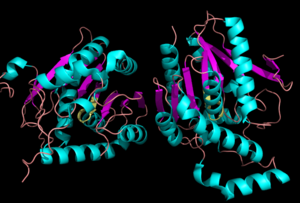
Figure 1:Crystal Structure of MGL Alpha helixes are in blue and beta sheets in purple. This protein is a dimer that is linked by antiparallel beta sheets
Background
Monoglyceride Lipase (MGL) is part of the α/β hydrolase family,a Serine hydrolase (Figure 1), having a [1]. MGL is present in most cells, providing the rate limiting step for the hydrolysis of monoacylglycerols (MG) into fatty acids and glycerol [2] . MGL also terminates the signaling of a primary endocannabinoid, 2-arachidonoyl glycerol (2-AG) [3]. MGL is the main enzyme responsible for hydrolyzing 2-arachidonoylglycerol into arachidonic acid and glycerol in vivo (Figure 3) [4]. One of the key features of MGL is the hydrophobic tunnel, which has been suggested to provide a model for drug research (Figure 7). [4]
Metabolic Role
Monoglyceride Lipase is involved in energy metabolism through two mechanisms. In the first mechanism of energy metabolism, MGL is able to hydrolyze monoacylglycerols into fatty acids and glycerol, which are able to then be used for energy production [2]. The second mechanism involves the degradation of 2-arachidonoyl glycerol (2-AG), which is a common endogenous ligand of cannabinoid receptors, by MGL. Looking in vivo of the metabolic role of MGL, MGL deficient mice showed increased monoacylglycerols in adipose tissue, brain, and liver as well as elevated 2-AG levels. With defective MGL, lipolysis is debilitated and diet-induced insulin resistance is reduced [2].
Component of Endocannabinoid System
MGL also degrades 2-Arachidonoylglycerol (2-AG), an endocannabinoid,
to a glycerol and arachidonic acid. [1] In the brain, endocannabinoids (ECs)are released from postsynaptic neurons, causing the retrograde suppression of synaptic transmission. [2]
In peripheral tissues, endocannabinoids (ECs) are active in autonomic nervous system. EC signalling affects processes such as learning, motor control, cognition, and pain [2]. EC signalling is also able to regulate lipid metabolism and food intake. Looking at the role of MGL in energy metabolism, a deficiency in MGL in animals led to the buildup of 2-AG [2]. The endocannabinoid 2-AG has a nociceptive effect in pain signalling [1]. MGL degrades 2-AG, preventing 2-AG from remaining bound to the cannabinoid receptor and therefore terminating the pain signal. Without the degradation of 2-AG by MGL, 2-AG levels would increase which would lead to a prolonged nociceptive effect [1]
Inhibition of MGL
The importance of Inhibition of Monoglyceride lipase is to keep it from breaking down 2-arachidonoyl glycerol. When 2-Ag is broken down it is not able to suppress pain and depression brain functions that human beings experience. N-arachidonyl maleimide (NAM) is one inhibitor of MGL that reacts with the amino acid . Figure 2
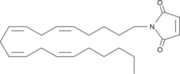
Figure 2: The structure of N-arachidonyl maleimide (NAM)that interacts with Cys252.
This Cysteine is buried in the active site near the catalytic serine and functions by sterically clashing with the natural ligand. A possible conformational change to Cys252 upon the binding of NAM could also lead to an inactive form of MGL.
MGL is also inhibited by being in complex with that is covalently bound to the catalytic Ser132. SAR629 adopts a Y shape and interacts with the MGL by hydrophobic interactions, with a few polar interactions as well. Figure 3
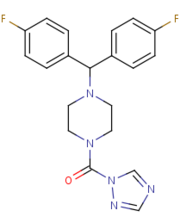
Figure 3: The structure and shape of SAR629.
With SAR629 interacting with the catalytic triad it inhibits the triad from breaking down 2-AG and inactivates MGL [4]
Structure
The of MGL has an eight-stranded β-sheet protein fold with seven parallel and one . Similar to the other α/β hydrolases, the β-sheets in the center of the protein surrounded by α-helices. The combination of the α-helices and β-sheets are able to provide a stable scaffold for the active site within MGL. MGL is monomeric. The catalytic triad, containing Ser132, His279, and Asp249, is found at the bottom of the binding pocket [4].
Catalytic triad
MGL has a classic that contains Ser-His-Asp. The triad was found using site-directed mutagenesis of each individual residue and each of these amino acid residues are catalytically essential to MGL [4]. The catalytic triad is located in the Binding Pocket buried at the bottom of it in the oxyanion hole connected by a water molecule.Figure 4
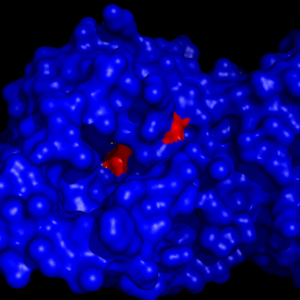
Figure 4: The binding pocket of MGL with the catalytic triad (shown in red) buried in it.
This triad has a natural attraction to Endocannabinoids, specifically 2-arachidonylglycerol (2-AG) [4].
Binding
2-AG binds to the catalytic triad and is hydrolyzed. The structure of 2-AG contains a long and flexible aliphatic chain and a polar head that is cleaved. It is the polar head that gets attracted to the catalytic triad and binds to it so the catalytic Serine can cleave 2-AG. 2-AG is broken down into arachidonic acid and glycerol which makes 2-AG inactive[4]. See Overall Reaction.
Inhibition of Catalytic Triad
Research on MGL is being geared towards inhibiting 2-AG from binding to the catalytic triad and being hydrolyzed. The binding of 2-AG to the catalytic triad can be extracted before being hydrolyzed. MPD (2-methyl-pentane-2,4-diol)is located at the end of the tunnel where the catalytic triad is at and the tunnel is filled with MPD molecules. MPD being in the same vicinity will extract 2-AG from the triad and the MPD molecule will sit in there in place of 2-AG. This is a natural inhibition phenomenon. Inhibition of MGL leads to increase in 2-AG levels since AG is broken down by MGL [1]. The catalytic triad is a major part of MGL and its interaction with other parts within the brain and how the brain functions.
Overall Reaction
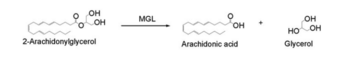
Figure 5: The breakdown of 2-AG into arachidonic acid +glyverol by MGL.
In this reaction 2-AG binds to the catalytic triad in the oxyanion hole in the active site. In the oxyanion holes the substrate is stabilized by two nitrogen atoms from the catalytic Histidine and Aspartate during the transition step of the catalytic reaction. The catalytic triad activates the nucleophilic serine and cleaves the ester bond of 2-AG that is being stabilized by its carbonyl group that is attached to the oxyanion hole. The glycerol molecule is released and it might diffuse to the narrow "exit hole", while the arachidonic acid would diffuse back to the top of the tunnel and leave the protein [4].
Ligand Binding Site
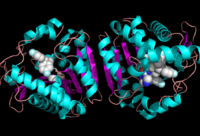
Figure 6: Ligand within the Overall Structure of MGL
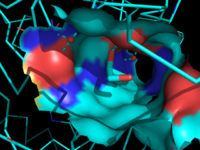
Figure 7: Ligand binding pocket showing the hydrophobic and polar regions
The of MGL has a large hydrophobic region with a polar bottom. The entrance of the binding pocket for MGL contains a lid, which is very flexible. The binding pocket or tunnel within MGL matches with the overall structure of 2-AG, with 2-AG's polar head being cleaved by the catalytic triad. Bertrand found that in MGL the binding pocket is not adjusted to the ligand's shape. However, the main movements of MGL associated with ligand binding involved the lid region. When 2-AG and its isomer 1(3)-AG bind to MGL, the hydrophobic chain is first aligned with the left part of the binding pocket. The carbonyl is then hydrogen bonded to . The polar head group of the ligand is then fixed by three hydrogen bonds. As a result, future research is looking into the large lipophilic portion of the binding pocket for designing selective inhibitors [4].
References
- ↑ 1.0 1.1 1.2 1.3 1.4 [Clemente, J. C., E. Nulton, M. Nelen, M. J. Todd, D. Maguire, C. Schalk-Hihi, L. C. Kuo, S.-P. Zhang, C. M. Flores, and J. K. Kranz. "Screening and Characterization of Human Monoglyceride Lipase Active Site Inhibitors Using Orthogonal Binding and Functional Assays." Journal of Biomolecular Screening 17.5 (2012): 629-40]
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 [Taschler, U., F. P. W. Radner, C. Heier, R. Schreiber, M. Schweiger, G. Schoiswohl, K. Preiss-Landl, D. Jaeger, B. Reiter, H. C. Koefeler, J. Wojciechowski, C. Theussl, J. M. Penninger, A. Lass, G. Haemmerle, R. Zechner, and R. Zimmermann. "Monoglyceride Lipase Deficiency in Mice Impairs Lipolysis and Attenuates Diet-induced Insulin Resistance." Journal of Biological Chemistry 286.20 (2011): 17467-7477]
- ↑ [Savinainen, Juha R., Megumi Yoshino, Anna Minkkilä, Tapio Nevalainen, and Jarmo T. Laitinen. "Characterization of Binding Properties of Monoglyceride Lipase Inhibitors by a Versatile Fluorescence-based Technique." Analytical Biochemistry 399.1 (2010): 132-34]
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 [ Bertrand, T., F. Augé, J. Houtmann, A. Rak, F. Vallée, V. Mikol, P.f. Berne, N. Michot, D. Cheuret, C. Hoornaert, and M. Mathieu. "Structural Basis for Human Monoglyceride Lipase Inhibition." Journal of Molecular Biology 396.3 (2010): 663-73.]







