This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Erythropoietin
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
<StructureSection load='1buy' size='450' side='right' scene=> | <StructureSection load='1buy' size='450' side='right' scene=> | ||
| - | You may include any references to papers as in: the use of JSmol in Proteopedia <ref>DOI 10.1007/BF02934992</ref> or to the article describing Jmol <ref>PMID: | + | You may include any references to papers as in: the use of JSmol in Proteopedia <ref>DOI 10.1007/BF02934992</ref> or to the article describing Jmol <ref>PMID: 9774108</ref> to the rescue. |
'''Intro''' | '''Intro''' | ||
| Line 11: | Line 11: | ||
'''History''' | '''History''' | ||
| - | Miyake et al first purified a few milligrams of erythropoietin from 2500 L human urine in 1977 | + | Miyake et al first purified a few milligrams of erythropoietin from 2500 L human urine in 1977 <ref>PMID: 6600633</ref>. This glycoprotein was first suspected as doctors observed anemia patients. Researchers noticed that patients with anemia had increased levels of erythropoiesis <ref>PMID:17253966</ref>. They noticed that the level of oxygen, seemed to be correlated with the amount of erythropoiesis, as well. After isolation, researchers worked to clone the gene so that they could produce the glycoprotein and treat hypoxia. The FDA approved this type of treatment in the 1990’s <ref>PMID:17253966</ref>. EPO works to stimulate the amount of red blood cells and the efficiency of hemoglobin, which increases oxygen library. This trait is what has led to many athletes to abuse this glycoprotein to improve their performance in their athletic sport, the most famous being Tour de France competitors (8). Most athletes involved in these scandal would intravenously inject themselves with this hormone (7). However, as medical techniques have advanced, some have started gene doping through in vivo and ex vivo gene transfer (7). |
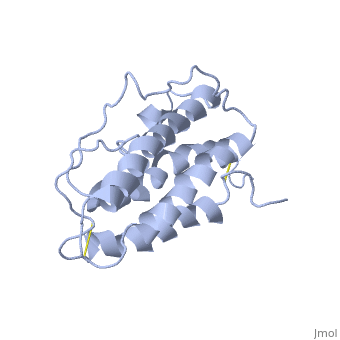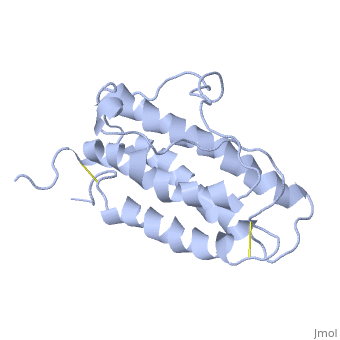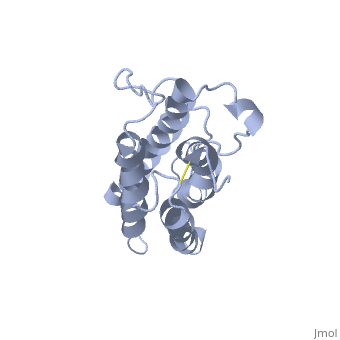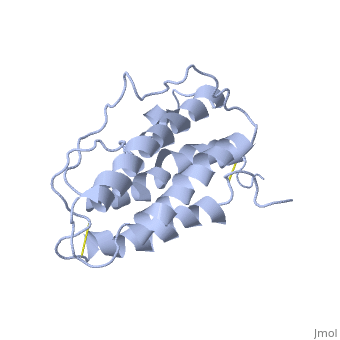
'''Structure'''. | '''Structure'''. | ||
| Line 24: | Line 24: | ||
''' | ''' | ||
| - | The <scene name='12/128258/Eporeceptor/3'>EPO receptor</scene> of the blood marrow is part of the hematipoietic cytokine family. This receptor has a single transmembrane domain, that forms a homodimer complex until it is activated by the binding of EPO.This receptor is 484 amino acids long and weigh 52.6 kDa <ref>PMID:17253966</ref>. Once the homodimer is formed after the binding, autophosphorlation of the Jak2 kinases, which activates other cellular processes <ref>DOI 10.1016/S1043-2760(98)00101-5</ref>. This transmembrane receptor has two extracellular domains. This receptor has two disulfide bonds that are formed from 4 cystine residues, <scene name='58/583377/Eporeceptord1d2cyslabel/1'>Cys67 and Cys83 and Cys28 and Cys38</scene> <ref>DOI 10.1016/S1043-2760(98)00101-5</ref>. The intracellular domain of this receptor does not possess any enzymatic activity like other receptors. When EPO comes in contact with the extracellular domains form a ligand bond. The extracellular sinding site 1 and Binding site 2 are composed of <scene name='58/583377/Eporeceptord1d2/1'>D1 and D2</scene> <ref>DOI 10.1016/S1043-2760(98)00101-5</ref>. When EPO binds, all loops on D1 and D2 of binding site one form a bind with EPO. However loop 4 of D1 on binding site 2 does not participate in the binding of EPO | + | The <scene name='12/128258/Eporeceptor/3'>EPO receptor</scene> of the blood marrow is part of the hematipoietic cytokine family. This receptor has a single transmembrane domain, that forms a homodimer complex until it is activated by the binding of EPO.This receptor is 484 amino acids long and weigh 52.6 kDa <ref>PMID:17253966</ref>. Once the homodimer is formed after the binding, autophosphorlation of the Jak2 kinases, which activates other cellular processes <ref>DOI 10.1016/S1043-2760(98)00101-5</ref>. This transmembrane receptor has two extracellular domains. This receptor has two disulfide bonds that are formed from 4 cystine residues, <scene name='58/583377/Eporeceptord1d2cyslabel/1'>Cys67 and Cys83 and Cys28 and Cys38</scene> <ref>DOI 10.1016/S1043-2760(98)00101-5</ref>. The intracellular domain of this receptor does not possess any enzymatic activity like other receptors. When EPO comes in contact with the extracellular domains form a ligand bond. The extracellular sinding site 1 and Binding site 2 are composed of <scene name='58/583377/Eporeceptord1d2/1'>D1 and D2</scene> <ref>DOI 10.1016/S1043-2760(98)00101-5</ref>. When EPO binds, all loops on D1 and D2 of binding site one form a bind with EPO. However loop 4 of D1 on binding site 2 does not participate in the binding of EPO <ref>PMID: 9774108</ref>. After the biniding of EPO, 8 tyrosine residues are phosphoralated which activates the <scene name='58/583377/Jak2/2'>Jak2 kinase</scene> <ref>DOI 10.1016/S1043-2760(98)00101-5</ref>. This kinase helps regulate the transcription of different genes and expression of other proteins. |
Revision as of 19:59, 24 April 2014
Erythropoietin Structure, Function, and History
| |||||||||||