This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1cza
From Proteopedia
| Line 1: | Line 1: | ||
| - | [[Image:1cza.gif|left|200px]] | + | [[Image:1cza.gif|left|200px]] |
| - | + | ||
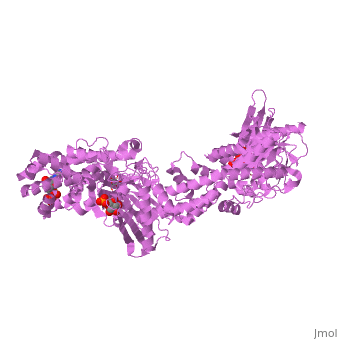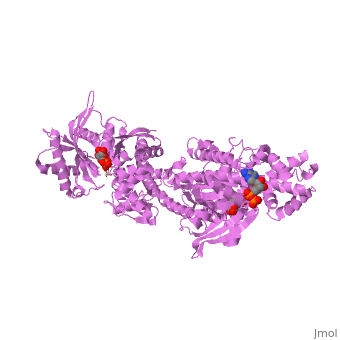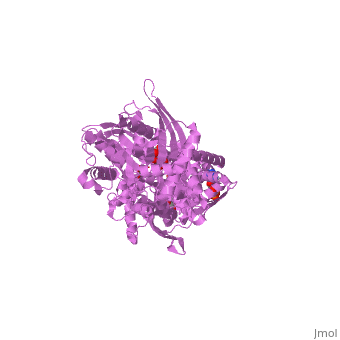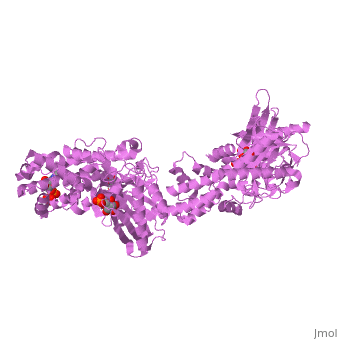
| - | '''MUTANT MONOMER OF RECOMBINANT HUMAN HEXOKINASE TYPE I COMPLEXED WITH GLUCOSE, GLUCOSE-6-PHOSPHATE, AND ADP''' | + | {{Structure |
| + | |PDB= 1cza |SIZE=350|CAPTION= <scene name='initialview01'>1cza</scene>, resolution 1.9Å | ||
| + | |SITE= | ||
| + | |LIGAND= <scene name='pdbligand=GLC:GLUCOSE'>GLC</scene>, <scene name='pdbligand=G6P:ALPHA-D-GLUCOSE-6-PHOSPHATE'>G6P</scene> and <scene name='pdbligand=ADP:ADENOSINE-5'-DIPHOSPHATE'>ADP</scene> | ||
| + | |ACTIVITY= [http://en.wikipedia.org/wiki/Hexokinase Hexokinase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=2.7.1.1 2.7.1.1] | ||
| + | |GENE= | ||
| + | }} | ||
| + | |||
| + | '''MUTANT MONOMER OF RECOMBINANT HUMAN HEXOKINASE TYPE I COMPLEXED WITH GLUCOSE, GLUCOSE-6-PHOSPHATE, AND ADP''' | ||
| + | |||
==Overview== | ==Overview== | ||
| Line 10: | Line 19: | ||
==About this Structure== | ==About this Structure== | ||
| - | 1CZA is a [ | + | 1CZA is a [[Single protein]] structure of sequence from [http://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1CZA OCA]. |
==Reference== | ==Reference== | ||
| - | Crystal structures of mutant monomeric hexokinase I reveal multiple ADP binding sites and conformational changes relevant to allosteric regulation., Aleshin AE, Kirby C, Liu X, Bourenkov GP, Bartunik HD, Fromm HJ, Honzatko RB, J Mol Biol. 2000 Mar 3;296(4):1001-15. PMID:[http:// | + | Crystal structures of mutant monomeric hexokinase I reveal multiple ADP binding sites and conformational changes relevant to allosteric regulation., Aleshin AE, Kirby C, Liu X, Bourenkov GP, Bartunik HD, Fromm HJ, Honzatko RB, J Mol Biol. 2000 Mar 3;296(4):1001-15. PMID:[http://www.ncbi.nlm.nih.gov/pubmed/10686099 10686099] |
[[Category: Hexokinase]] | [[Category: Hexokinase]] | ||
[[Category: Homo sapiens]] | [[Category: Homo sapiens]] | ||
| Line 27: | Line 36: | ||
[[Category: G6P]] | [[Category: G6P]] | ||
[[Category: GLC]] | [[Category: GLC]] | ||
| - | [[Category: structurally homologous | + | [[Category: structurally homologous domain]] |
| - | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Mar 20 10:30:46 2008'' |
Revision as of 08:30, 20 March 2008
| |||||||
| , resolution 1.9Å | |||||||
|---|---|---|---|---|---|---|---|
| Ligands: | , and | ||||||
| Activity: | Hexokinase, with EC number 2.7.1.1 | ||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||
MUTANT MONOMER OF RECOMBINANT HUMAN HEXOKINASE TYPE I COMPLEXED WITH GLUCOSE, GLUCOSE-6-PHOSPHATE, AND ADP
Contents |
Overview
Hexokinase I, the pacemaker of glycolysis in brain tissue, is composed of two structurally similar halves connected by an alpha-helix. The enzyme dimerizes at elevated protein concentrations in solution and in crystal structures; however, almost all published data reflect the properties of a hexokinase I monomer in solution. Crystal structures of mutant forms of recombinant human hexokinase I, presented here, reveal the enzyme monomer for the first time. The mutant hexokinases bind both glucose 6-phosphate and glucose with high affinity to their N and C-terminal halves, and ADP, also with high affinity, to a site near the N terminus of the polypeptide chain. Exposure of the monomer crystals to ADP in the complete absence of glucose 6-phosphate reveals a second binding site for adenine nucleotides at the putative active site (C-half), with conformational changes extending 15 A to the contact interface between the N and C-halves. The structures reveal distinct conformational states for the C-half and a rigid-body rotation of the N-half, as possible elements of a structure-based mechanism for allosteric regulation of catalysis.
Disease
Known disease associated with this structure: Hemolytic anemia due to hexokinase deficiency OMIM:[142600]
About this Structure
1CZA is a Single protein structure of sequence from Homo sapiens. Full crystallographic information is available from OCA.
Reference
Crystal structures of mutant monomeric hexokinase I reveal multiple ADP binding sites and conformational changes relevant to allosteric regulation., Aleshin AE, Kirby C, Liu X, Bourenkov GP, Bartunik HD, Fromm HJ, Honzatko RB, J Mol Biol. 2000 Mar 3;296(4):1001-15. PMID:10686099
Page seeded by OCA on Thu Mar 20 10:30:46 2008