This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox 126
From Proteopedia
(Difference between revisions)
| Line 5: | Line 5: | ||
==='''Background Information'''=== | ==='''Background Information'''=== | ||
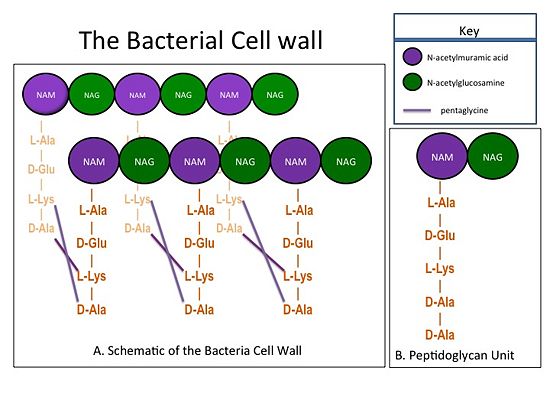
| - | The bacterial cell wall is composed of sheets of peptidoglycan cross-linked together to form a highly polymeric "mesh" that helps maintain the structural strength of the cell (Figure 1). A peptidoglycan sheet consists of alternating residues of <font color='purple'> '''N-acetylmuramic acid (NAM)''' </font> and <font color='green'> '''N-acetylglucosamine (NAG)''' </font> linked together by β-(1,4)- glycosidic bonds. In ''Staphylococcus aureus'' (S. aureus), the NAM residues are coupled to a (D-Ala) residues. The sheets of peptidoglycan are cross-linked together with pentaglycine chains. The cross-linking of adjacent peptidoglycan sheets is catalyzed by transpeptidases (TP). Beta-Lactam antibiotics, such as penicillin and the anti-MRSA cephlosporins, ceftobiprole and ceftaroline, stop the production of the cell wall, and so kill bacteria, by irreversibly inhibiting TPs. Therefore, TPs are also called penicillin-binding proteins.[[Image:CellWall.jpg|thumb|alt= Alt text| Figure 1.(A) This moiety is polymerized to form sheets of peptidoglycan. Adjacent sheets of peptidoglycan are cross-linked together by pentaglycine "bridges" to form a polymeric "mesh" that is essential for the structural integrity of the bacterial cell (B) The cell wall is composed of repeating units of a NAM/NAG disaccharide and peptide moiety; ''i.e.'', peptidoglycan |550px]] | + | The bacterial cell wall is composed of sheets of peptidoglycan cross-linked together to form a highly polymeric "mesh" that helps maintain the structural strength of the cell (Figure 1). A peptidoglycan sheet consists of alternating residues of <font color='purple'> '''N-acetylmuramic acid (NAM)''' </font> and <font color='green'> '''N-acetylglucosamine (NAG)''' </font> linked together by β-(1,4)- glycosidic bonds. In ''Staphylococcus aureus'' (S. aureus), the NAM residues are coupled to a (D-Ala) residues. The sheets of peptidoglycan are cross-linked together with pentaglycine chains. The cross-linking of adjacent peptidoglycan sheets is catalyzed by transpeptidases (TP). Beta-Lactam antibiotics, such as penicillin and the anti-MRSA cephlosporins, ceftobiprole and ceftaroline, stop the production of the cell wall, and so kill bacteria, by irreversibly inhibiting TPs. Therefore, TPs are also called penicillin-binding proteins.[[Image:CellWall.jpg|thumb|alt= Alt text| Figure 1.(A) This moiety is polymerized to form sheets of peptidoglycan. Adjacent sheets of peptidoglycan are cross-linked together by pentaglycine "bridges" to form a polymeric "mesh" that is essential for the structural integrity of the bacterial cell. (B) The cell wall is composed of repeating units of a NAM/NAG disaccharide and peptide moiety; ''i.e.'', peptidoglycan. |550px]] |
| Line 11: | Line 11: | ||
==='''Mechanism of action of Beta-Lactam Antibiotics'''=== | ==='''Mechanism of action of Beta-Lactam Antibiotics'''=== | ||
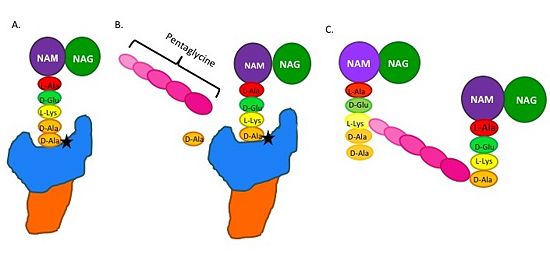
| - | The beta-lactam antibiotics irreversibly bind to and inhibit TPs. This results in the disruption of peptidoglycan synthesis and ultimately cell growth. Specifically, beta-lactams, such as penicillin and the anti-MRSA cephlasporins, ceftobiprole and ceftaroline, are molecular mimics of the peptidoglycan D-Ala-D-Ala moiety; the normal TP substrate (Figure 2; Tipper and Strominger, 1965). Therefore, they "trick" the TP active site serine residue to react with them, resulting in the irreversible inhibition of TP activity and of cell wall synthesis.[[Image: MechanismofPBP.jpg|thumb|alt= Alt text|Figure 2. Schematic showing Catalytic Mechanism of PBP2a (A) the peptidoglycan |550px]] | + | The beta-lactam antibiotics irreversibly bind to and inhibit TPs. This results in the disruption of peptidoglycan synthesis and ultimately cell growth. Specifically, beta-lactams, such as penicillin and the anti-MRSA cephlasporins, ceftobiprole and ceftaroline, are molecular mimics of the peptidoglycan D-Ala-D-Ala moiety; the normal TP substrate (Figure 2; Tipper and Strominger, 1965). Therefore, they "trick" the TP active site serine residue to react with them, resulting in the irreversible inhibition of TP activity and of cell wall synthesis.[[Image: MechanismofPBP.jpg|thumb|alt= Alt text|Figure 2.Schematic showing Catalytic Mechanism of PBP2a (A) The peptidoglycan D-Ala D-Ala moiety enters the TP active site, which is in the TP domain (blue) (B) The active site serine residue (star) reacts with and breaks the peptide bond between the D-Ala residues. The terminal D-Ala residue exits the active site. The remaining D-Ala residue is covalently bound to the active site serine residue, and therefore, to TP. The incoming pentaglycine chain reacts with the bound D-Ala residue and is cross-linked to the D-Ala residue. (D) This results in cross-linking between adjacent peptidoglycan "sheets" and regeneration of the active site serine residue so it can catalyze another cross-linking reaction. |550px]] |
==='''MRSA, PBP2a, and anti-MRSA Cephalosporins'''=== | ==='''MRSA, PBP2a, and anti-MRSA Cephalosporins'''=== | ||
Revision as of 18:20, 31 July 2014
| |||||||||||